Advertisements
Advertisements
प्रश्न
How are nonstoichiometric point defects classified? Explain with diagram the metal deficiency defect.
उत्तर
- Nonstoichiometric point defects are classified into following two types:
- Metal deficiency defect
- Metal excess defect
- Metal deficiency defect:
- This defect is possible only in compounds of metals that show variable oxidation states.
- In some crystals, positive metal ions are missing from their original lattice sites. The extra negative charge is balanced by the presence of cation of the same metal with higher oxidation state than that of missing cation.
e.g.: In the compound NiO, one Ni2+ ion is missing creating a vacancy at its lattice site. The deficiency of two positive charges is made up by the presence of two Ni3+ ions at the other lattice sites of Ni2+ ions. The composition of NiO then becomes Ni0.97O1.0.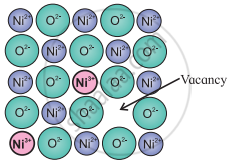
APPEARS IN
संबंधित प्रश्न
What are the consequences of Schottky defect?
What is an impurity defect? What are its types? Explain the formation of vacancies through aliovalent impurity with example.
Give the classification of stoichiometric point defects.
Which of the following is an example of substitutional impurity defect?
Which of the following match is CORRECT?
(I) Frenkel defect: Electrical neutrality of the compound is preserved.
(II) Schottky defect: Density of the substance decreases.
(III) Schottky defect: Combination of vacancy defect and interstitial defect.
(IV) Frenkel defect: Small difference between size of cation and anion.
Which among the following statements is true about Schottky defect?
Which of the following pair of ionic crystals show Schottky defect?
Schottky defect in crystals is observed when ____________.
Schottky defect is formed in ionic compounds with ____________.
Write the consequences of Schottky defect with reasons.
Explain metal deficiency defect with examples.
AgBr shows which type of defect?
Name a compound where Frenkel defect is found.
In which among the following solids, Schottky defect is not observed?
When electrons are trapped into the crystal in anion vacancy, the defect is known as ______.
Explain the following term:
Substitutional impurity defect
Explain the following term:
Interstitial impurity defect
Explain Self interstitial defect in elemental solid.
What is a crystal defect?
Give the disadvantages of a vacancy defect.
