Advertisements
Advertisements
प्रश्न
How is copper extracted from its sulphide ore (copper glance), Cu2S? Explain with equations of the reactions involved.
उत्तर
The sulphide ore of copper is copper glance. The compound present in the ore is copper (I) sulphide. Its formula is Cu2S. Copper is a less reactive metal; therefore, it is placed at the bottom in the reactivity series. Copper is extracted by heating its sulphide ore alone.
Two steps involved in the extraction of copper metal from concentrated copper glance ore are:
1. Roasting: Copper glance, being a sulphide ore, is heated in the presence of air. Copper (I) sulphide is converted to copper (I) oxide.
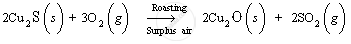
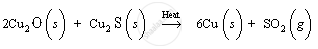
Copper (I) oxide, so formed, reacts with non-oxidised copper (I) sulphide ore in the absence of air to form copper metal. When the mixture of Cu2O and Cu2S is heated, they react together to give copper metal and sulphur dioxide gas.
APPEARS IN
संबंधित प्रश्न
Fill in the following blank with suitable word:
The rocky material found with ores is called.............
An ore of manganese metal is:
(a) bauxite
(b) haematite
(c) cuprite
(d) pyrolusite
Which of the following metals can be obtained from haematite ore?
(a) copper
(b) sodium
(c) zinc
(d) iron
Name the following:
Two metals which are found in the free or native state.
Define the term : Gangue
How an ore is concentrated by froth floatation process?
The following questions relate to the extraction of aluminium by electrolysis.
Explain why it is necessary to renew the anode periodically.
Compare the properties of a typical metal and a non- metal on the basis of the following:
(a) electronic configuration
(b) nature of oxides
(c) oxidizing or reducing action
(d) conductivity of heat and electricity
Observe the given figure of reactivity series of metals and answer the following questions:

Reactivity series of metals
- Name two metals which react with water.
- Name two moderately reactive metals.
- Name the most highly reactive metal and the most less reactive metal.
Answer the questions on the following passage.
The minerals from which the metal can be separated economically are called ores. Ores contain many types of impurities such as soil, sand and rocky substances along with metal compounds. These impurities are called gangue.
Metals can be extracted from their ores by means of various methods of separation. The process of extraction of metal in a pure state from the ores is also a part of metallurgy.
Ores are taken out from the mines and the gangue is usually separated from the ore at the site itself by various methods. Then the ores are carried out to the place where metals are produced. Here metals are extracted in pure form. Then metals are further purified by different methods of purification. This entire process is called metallurgy. Most metals being reactive do not occur in nature in the free state but are found in combined state as their salts such as oxides, carbonates, sulphides, and nitrates. however, the most unreactive metals that are not affected by air, water and other natural factors like silver, gold, platinum, generally occur in a free state. The compounds of metals that occur in nature along with the impurities are called minerals.
- What are ores?
- Which processes are involved in the branch of metallurgy? What is metallurgy?
- Which metals are found in a free state?
- In what forms are metals found in combined state?
- What is gangue?
