Advertisements
Advertisements
प्रश्न
How is copper extracted from its sulphide ore (copper glance), Cu2S? Explain with equations of the reactions involved.
उत्तर
The sulphide ore of copper is copper glance. The compound present in the ore is copper (I) sulphide. Its formula is Cu2S. Copper is a less reactive metal; therefore, it is placed at the bottom in the reactivity series. Copper is extracted by heating its sulphide ore alone.
Two steps involved in the extraction of copper metal from concentrated copper glance ore are:
1. Roasting: Copper glance, being a sulphide ore, is heated in the presence of air. Copper (I) sulphide is converted to copper (I) oxide.
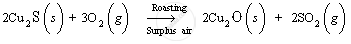
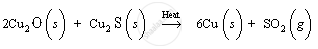
Copper (I) oxide, so formed, reacts with non-oxidised copper (I) sulphide ore in the absence of air to form copper metal. When the mixture of Cu2O and Cu2S is heated, they react together to give copper metal and sulphur dioxide gas.
APPEARS IN
संबंधित प्रश्न
In the extraction of aluminium: Name the process of concentration of bauxite.
In the extraction of aluminium Write the cathode reaction in electrolytic reduction of alumina.
How is manganese extracted from manganese dioxide, MnO2? Explain with the help of an equation.
Explain how, mercury is extracted from its sulphide ore (cinnabar). Give equations of the reactions involved.
Name the following:
Two metals which are found in the free or native state.
How an ore is concentrated by froth floatation process?
Mercury, silver, gold are highly reactive metals.
Gold and silver are active metals.
Explain the characteristics of ionic compounds.
Which of the statements about the reaction, \[\ce{ZnO + CO -> Zn + CO2}\] is correct?
