Advertisements
Advertisements
प्रश्न
How an ore is concentrated by froth floatation process?
उत्तर
Concentrated of an ore by froth floatation process : This process depends on preferential wettability of the ore and the gangue particles. Crushed ore is taken in
a large tank containing water and certain oils. The ore particles get wetted by the oil and the gangue particles get wetted by water. The mixture is then agitated with
the help of compressed air. The ore particles that get wetted with the oil form a froth on the top, and can be scooped out. This method is used for the concentration
of sulphide ores.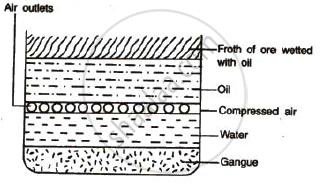
Figure : Froth floatation process
APPEARS IN
संबंधित प्रश्न
Fill in the following blank with suitable word:
The rocky material found with ores is called.............
What is the difference between a mineral and an ore?
Explain how, mercury is extracted from its sulphide ore (cinnabar). Give equations of the reactions involved.
In an electrolytic tank, aluminium metal is being extracted by the electrolysis of molten aluminium oxide using carbon electrodes. It is observed that one of the carbon electrodes is gradually burnt away and has to be replaced.
(a) Which carbon electrode (cathode or anode) is burnt away?
(b) Why is this carbon electrode burnt away?
An element has its electron configuration as 2,8,8,2. Now answer the following questions.
a) What is the atomic number of this element?
b) What is the group of this element?
c) To which period does this element belong?
Define the following term.
Gangue
Give the chemical formula of :
Sodium aluminate
Name the following:
The process of heating a substance very strongly in such a way that it does not combine with oxygen.
To protect iron from rusting, it is coated with a thin layer of zinc. Name the process.
The given reaction shows one of the processes to extract the metals like Iron and Manganese.
\[\ce{MnO2(s) + Al(s) -> Mn(l) + Al2O3(s) + Heat}\]
Identify the substance oxidised and reduced in the above reaction.
