Advertisements
Advertisements
Question
How an ore is concentrated by froth floatation process?
Solution
Concentrated of an ore by froth floatation process : This process depends on preferential wettability of the ore and the gangue particles. Crushed ore is taken in
a large tank containing water and certain oils. The ore particles get wetted by the oil and the gangue particles get wetted by water. The mixture is then agitated with
the help of compressed air. The ore particles that get wetted with the oil form a froth on the top, and can be scooped out. This method is used for the concentration
of sulphide ores.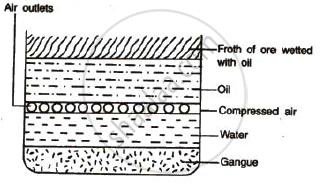
Figure : Froth floatation process
APPEARS IN
RELATED QUESTIONS
The process by which sulphide ore is concentrated.
In the extraction of aluminium: Why is it necessary to replace anodes time to time?
Name one metal which is extracted by reduction with heat alone.
Which metal is extracted from cinnabar ore?
Which of the following metals can be obtained from haematite ore?
(a) copper
(b) sodium
(c) zinc
(d) iron
A sulphide ore is converted into metal oxide by the process of:
(a) carbonation
(b) roasting
(c) calcination
(d) anodising
Metals X and Y can be recovered from the anode mud left behind after the electrolytic refining of copper metal. The coins made of metal X look new even after several years of use but the coins made of metal Y lose their shine gradually and get blackened soon. When metal X is alloyed with a small amount of metal Y, it becomes hard and hence suitable for making ornaments. What are metals X and Y? Also state the colour of metal X.
Name the following:
The process in which an ore is heated in air so that oxygen gets added to it to form the oxides.
_______ is the least reactive metal.
Write the name.
The molecular formula of main ore of aluminium –
