Advertisements
Advertisements
प्रश्न
How does perspiration or sweating help keep our body cool on a hot day ?
उत्तर
High temperature in the surroundings rises our body temperature. This rise in the body temperature is accompanied with secretion of sweat from the sweat glands. This sweat absorbs the body heat to achieve its latent heat of vaporization and evaporates. In the process, body temperature decreases due to release of body heat to sweat.
APPEARS IN
संबंधित प्रश्न
Convert the following temperature to the celsius scale.
293 K
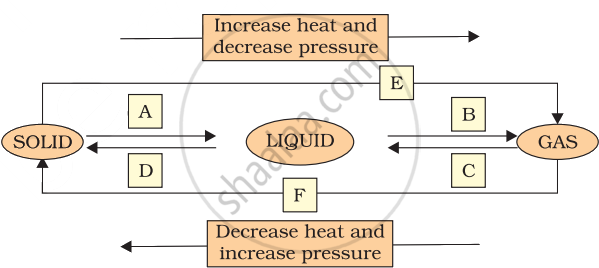
Name A, B, C, D, E and F in the following diagram showing change in its state.
How do you say that the process of digestion is a chemical change ?
Convert the temperature of 373° C to the Kelvin scale.
What is meant by saying that the latent heat of vaporisation of water is 22.5 × 105 J/kg ?
State whether the following statement is true or false :
Solid carbon dioxide is stored under low pressure.
When water is cooled to a temperature x, it gets converted into ice at temperature x by a process called P. And when ice at temperature x is warmed, it gets reconverted into water at the same temperature x in a process called Q.
- What is the value of temperature x in Kelvin ?
- What is the process P known as ?
- What is the name of energy released during process P ?
- What is the process Q known as ?
- What is the name of energy absorbed during process Q ?
Define: cohesive force
Fill in the blanks:
The molecules are at a greater distance in ______ compared to liquids.
State the property of the following substance.
Lead chloride reacts with barium sulphate to give a white precipitate of lead sulphate.
