Advertisements
Advertisements
प्रश्न
How will you bring about the following conversion?
But-1-ene to but-2-ene
उत्तर
\[\begin{array}{cc}
\ce{\underset{But-1-ene}{CH3CH2CH = CH2} ->[HBr][Markonikoff's rule] CH3 - CH2 - CH - CH3 ->[Alc. KOH, \Delta][-HBr] \underset{But-2-ene}{CH3 - CH = CH - CH3}}\\
\phantom{....}|\\
\phantom{.....}\ce{\underset{2-Bromobutane}{Br}}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Identify ‘A’ and ‘B’ in the following reaction :
\[\ce{CH3 - CH = CH2 ->[HBr]'A' ->[alc.KOH]'B'}\]
How do you convert: 2-bromobutane to but-2-ene
Predict all the alkenes that would be formed by dehydrohalogenation of the following halide with sodium ethoxide in ethanol and identify the major alkene:
2, 2, 3-Trimethyl-3-bromopentane
Write the structure of the major organic product in the following reaction:
\[\ce{(CH3)3CBr + KOH ->[ethanol][heat]}\]
How the following conversion can be carried out?
1-Bromopropane to 2-bromopropane
How the following conversion can be carried out?
2-Chloropropane to 1-propanol
How the following conversion can be carried out?
2-Bromopropane to 1-bromopropane
Draw a neat, labelled energy profile diagram for SN1 reaction mechanism.
Observe the following compounds and answer the questions given below.
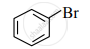
(I)
\[\ce{\underset{\text{(II)}}{CH3 - CH2 - Br}}\]
- Identify the type of halides.
- Explain the nature of the C – Br bond in both of these halides.
- Which of these compounds will undergo aqueous alkaline hydrolysis readily? Write the reaction in support of your answer.

'A' is:
Deamination of meso- di bromobutane gives mainly:-
Identify the major product formed when 2-cyclohexylchloroethane undergoes a dehydrohalogenation reaction. Name the reagent which is used to carry out the reaction.
Elimination of bromine from 2-bromobutane results in the formation of ______.
The conversion of an alkyl halide into an alkene by alcoholic KOH is classified as ______.
