Advertisements
Advertisements
प्रश्न
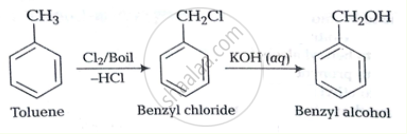
How will you bring about the following conversion?
Toluene to benzyl alcohol
How the following conversion can be carried out?
Toluene to benzyl alcohol
उत्तर
APPEARS IN
संबंधित प्रश्न
Write the structure of an isomer of compound C4H9Br which is most reactive towards SN1 reaction
Write the structures of A, B and C in the following:

Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
CH3CH2CH2CH2Br or \[\begin{array}{cc}
\ce{CH3CH2CHCH3}\\
\phantom{...}|\\
\phantom{....}\ce{Br}\
\end{array}\]
Write the isomers of the compound having the formula C4H9Br.
What are ambident nucleophiles? Explain with an example.
What is the action of the following on ethyl bromide
alcoholic solution of potassium hydroxide.
In a coordination entity of the type [PtCl2(en)2]2+ which isomer will show optical isomerism?
Which of the following reactions is an example of nucleophilic substitution reaction?
Which of the following undergoes nucleophilic substitution exclusively by SN1 mechanism?
A primary alkyl halide would prefer to undergo ______.
Aryl halides are extremely less reactive towards nucleophilic substitution. Predict and explain the order of reactivity of the following compounds towards nucleophilic substitution:
| (I) | 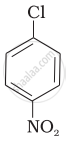 |
| (II) | 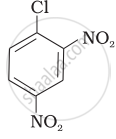 |
| (III) | 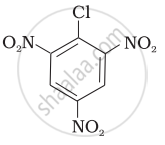 |
Which one of the following compounds is more reactive towards SN1 reaction?
In which reaction mechanism carbocation is formed?
Among the following compounds I - IV, which one forms a yellow precipitate on reacting sequentially with (i) NaOH (ii) dil. HNO3 (iii) AgNO3?
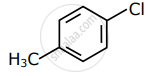 |
 |
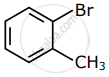 |
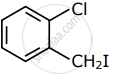 |
| I | II | III | IV |
Which of the following compounds will show retention in configuration on nucleophile substitution by OH− ion?
The number of chiral carbons present in the molecule given below is ______.
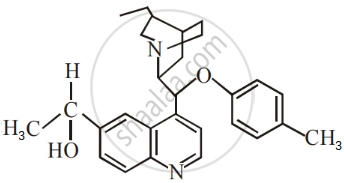
The decreasing order of reactivity of the following compounds towards nucleophilic substitution (SN2) is ______.
Arrange the following compounds in increasing order of reactivity towards SN2 reaction.
2-Bromopentane, 1-Bromopentane, 2-Bromo-2-methylbutane