Advertisements
Advertisements
प्रश्न
How would you show that hydrogen is a non-supporter of combustion?
उत्तर
Hold a hydrogen gas filled jar with its mouth downwards.
Place a lighted candle inside the jar. The candle gets extinguished but the gas bums with a pop sound. This shows that hydrogen is non-supporter of combustion.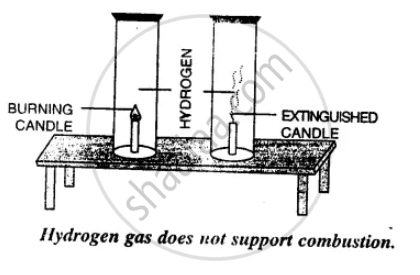
APPEARS IN
संबंधित प्रश्न
Name the chemicals required to prepare hydrogen gas in the laboratory.
FILL IN THE BLANK
........................ zinc is preferred over pure zinc in the laboratory preparation of hydrogen.
Starting from zinc how would you obtain hydrogen using Steam.
[Give balanced equation & name the product formed in the case other than hydrogen].
Name a metal which will not react with the reactants above to give hydrogen.
Starting from zinc how would you obtain hydrogen using an alkali.
[Give balanced equation & name the product formed in the case other than hydrogen].
Name a metal which will not react with the reactants above to give hydrogen.
Give balanced equation for the following conversion:
Acidified water to hydrogen – by electrolysis.
Give reason for the following:
Copper does not displace hydrogen from dilute hydrochloric acid, but zinc does.
In the laboratory preparation of hydrogen from zinc and dil. acid. Give a reason for the following:
The complete apparatus is air-tight.
In the laboratory preparation of hydrogen from zinc and dil. acid. Give a reason for the following:
Dilute nitric acid is not preferred as the reactant acid.
Give a reason for the following.
Nitric acid in the dilute form is not used in the laboratory preparation of hydrogen from metals.
The diagram represent the preparation and collection of hydrogen by a standard laboratory method.

State what difference will be seen if pure zinc is added in the distillation flask ‘X’ instead of granulated zinc.
