Advertisements
Advertisements
प्रश्न
Identify the metal and justify your answer.
Carbonyl \[\ce{M(CO)5}\]
उत्तर
\[\ce{Fe(CO)5}\] as per EAN Rule.
EAN Rule: Effective atomic number (EAN), number that represents the total number of electrons surrounding the nucleus of a metal atom in a metal complex.
APPEARS IN
संबंधित प्रश्न
Why do interstitial compounds have higher melting points than corresponding pure metals?
Explain briefly how +2 state becomes more and more stable in the first half of the first row transition elements with increasing atomic number?
For M2+/M and M3+/M2+ systems, the EΘ values for some metals are as follows:
| Cr2+/Cr | −0.9 V |
| Mn2+/Mn | −1.2 V |
| Fe2+/Fe | −0.4 V |
| Cr3/Cr2+ | −0.4 V |
| Mn3+/Mn2+ | +1.5 V |
| Fe3+/Fe2+ | +0.8 V |
Use this data to comment upon:
The stability of Fe3+ in acid solution as compared to that of Cr3+ or Mn3+.
Give reasons: E° value for the Mn3+/Mn2+ couple is much more positive than that for Fe3+/Fe2+.
Why does the density of transition elements increase from Titanium to Copper? (at. no. Ti = 22,
Cu = 29)
Although \[\ce{Cr^3+}\] and \[\ce{Co^2+}\] ions have same number of unpaired electrons but the magnetic moment of \[\ce{Cr^3+}\] is 3.87 B.M. and that of \[\ce{Co^2+}\] is 4.87 B.M. Why?
Match the properties given in Column I with the metals given in Column II.
| Column I (Property) | Column II (Metal) | |
| (i) | An element which can show +8 oxidation state | (a) \[\ce{Mn}\] |
| (ii) | 3d block element that can show | (b) \[\ce{Cr}\] |
| upto +7 oxidation state | (c) \[\ce{Os}\] | |
| (iii) | 3d block element with highest melting point | (d) \[\ce{Fe}\] |
The trend of which property is represented by the following graph?
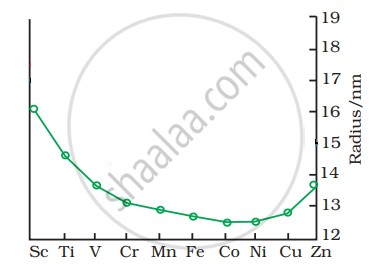
A pair of coloured ions is ______.
A coordination compound has the formula \[\ce{CoCl3.4NH3}\]. It precipitates silver ions as AgCl and its molar conductance corresponds to a total of two ions.
Based on this information, answer the following question:
- Deduce the structural formula of the complex compound.
- Write the IUPAC name of the complex compound.
- Draw the geometrical isomers of the complex compound.
