Advertisements
Advertisements
प्रश्न
If you take some distilled water in a test-tube, add an equal amount of acetic acid to it, shake the test-tube well and leave it undisturbed on the test-tube stand, then after about 5 minutes what would you observe?
(A) There is a layer of water over the layer of acetic acid.
(B) A precipitate is setting at the bottom of the test-tube.
(C) Bubbles of colourless gas are coming out of the test-tube.
(D) There is a clear, colourless transparent solutions in the test-tube.
उत्तर
Acetic acid is completely soluble in distilled water. Therefore, if we add equal amount of acetic acid in a test-tube containing distilled water, then there will be a clear, colourless transparent solutions in the test-tube observe.
Hence, the correct answer is option (D).
APPEARS IN
संबंधित प्रश्न
Metal compound A reacts with dilute hydrochloric acid to produce effervescence. The gas evolved extinguishes a burning candle. Write a balanced chemical equation for the reaction if one of the compounds formed is calcium chloride.
Complete and balance the following chemical equations:
CuO (s) + HCI (aq) →
On adding dilute hydrochloric acid to copper oxide powder, the solution formed is blue-green.
Predict the new compound formed which imparts a blue-green colour to solution.
Hydrochloric acid reacts with a metal X to form a gas Y which burns with a 'pop' sound. Sodium hydroxide solution also reacts with the same metal X (on heating) to form the same gas Y.
Write the chemical equation of the reaction of metal X with (i) hydrochloric acid, and (ii) sodium hydroxide solution.
With which substance should chlorine be treated to get bleaching powder?
When _______________ is passed through fresh lime water, it turns milky.
Acids turn blue litmus paper to ______.
The table given below shows the reaction of a few elements with acids and bases to evolve Hydrogen gas.
| Element | Acid | Base |
| A |
✗
|
✗ |
| B |
✓
|
✓ |
| C | ✓ | ✗ |
| D | ✓ | ✓ |
Which of these elements form amphoteric oxides?
Which of the following gives the correct increasing order of acidic strength?
In an attempt to demonstrate electrical conductivity through an electrolyte, the apparatus setup. Which among the following statement(s) is(are) correct?
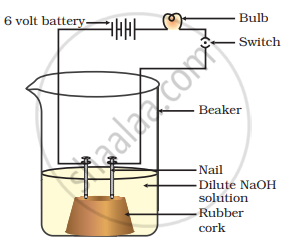
- Bulb will not glow because electrolyte is not acidic
- Bulb will glow because NaOH is a strong base and furnishes ions for conduction.
- Bulb will not glow because circuit is incomplete
- Bulb will not glow because it depends upon the type of electrolytic solution
