Advertisements
Advertisements
प्रश्न
Mark the correct options.
(a) An atom with a vacancy has smaller energy that a neutral atom.
(b) K X-ray is emitted when a hole makes a jump from the K shell to some other shell.
(c) The wavelength of K X-ray is smaller than the wavelength of L X-ray of the same material.
(d) The wavelength of Kα X-ray is smaller than the wavelength of Kβ X-ray of the same material.
उत्तर
(b) K X-ray is emitted when a hole makes a jump from the K shell to some other shell.
(c) The wavelength of K X-ray is smaller than the wavelength of L X-ray of the same material.
Energy of a vacant atom is higher than that of a neutral atom.
Hence, option (a) is incorrect.
K X-ray is emitted when an electron makes a jump to the K shell from some other shell. As a result, a positive charge hole is created in the outer shell. As the electron continuously moves to the K shell, the hole moves from the K shell to some other shell. Hence, option (b) is correct.
K X-ray is emitted due to the transition of an electron from the L or M shell to the K shell and L X-ray is emitted due to the transition of an electron from the M or N shell to the L shell. The energy involved in the transition from the L or M shell to the K shell is higher than the energy involved in the transition from the M or N shell to the L shell. Since the energy is inversely proportional to the wavelength, the wavelength of the K X-ray is smaller than the wavelength of the L X-ray of the same material. Hence, option (c) is correct.
If EK, EL and EM are the energies of K, L and M shells, respectively, then the wavelength of Kα X-ray (`lambda_1`) is given by
`lambda_1 = (hc)/(E_K - E_L)`
Here,
h = Planck's constant
c = Speed of light
Wavelength of the Kβ X-ray (`lambda_2`) is given by
`lambda_2 = (hc)/(E_K - E_M)`
As the difference of energies (`E_K - E_M`) is more than `(E_K - E_L)`, `λ_2` is less than `λ_1` Hence, option (d) is not correct.
APPEARS IN
संबंधित प्रश्न
If the earth did not have atmosphere, would its average surface temperature be higher or lower than what it is now? Explain.
To which part of electromagnetic spectrum does a wave of frequency 3 × 1013 Hz belong?
Give a reason for the following:
It is necessary to use satellites for long-distance TV transmission. Why?
A wave has a wavelength of 10-3 nm. Name the wave.
What do you understand by the invisible spectrum?
An electromagnetic wave has a frequency of 500 MHz and a wavelength of 60 cm Calculate the velocity of the wave.
In a Coolidge tube, electrons strike the target and stop inside it. Does the target get more and more negatively charged as time passes?
Can X-rays be used for photoelectric effect?
What potential difference should be applied across an X-ray tube to get X-ray of wavelength not less than 0.10 nm? What is the maximum energy of a photon of this X-ray in joule?
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
The Kβ X-rays from certain elements are given below. Draw a Moseley-type plot of √v versus Z for Kβ radiation.
| Element | Ne | P | Ca | Mn | Zn | Br |
| Energy (keV) | 0.858 | 2.14 | 4.02 | 6.51 | 9.57 | 13.3 |
Heat at the rate of 200 W is produced in an X-ray tube operating at 20 kV. Find the current in the circuit. Assume that only a small fraction of the kinetic energy of electrons is converted into X-rays.
Name the scientist who discovered radio waves
Name three radiations and their wavelength range which are invisible and beyond the violet end of the visible spectrum.
Answer the following question.
Gamma rays and radio waves travel with the same velocity in free space. Distinguish between them in terms of their origin and the main application.
State three properties of infrared radiations similar to that of visible light.
Give one use of electromagnetic radiation in Ultraviolet radiation.
The ozone layer absorbs
The electric field intensity produced by the radiations coming from 100 W bulb at a 3 m distance is E. The electric field intensity produced by the radiations coming from 50 W bulb at the same distance is ______.
Which is the correct ascending order of wavelengths?
In an atom X, electrons absorb the energy from an external source. This energy “excites” the electrons from a lower-energy level to a higher-energy level around the nucleus of the atom. When electrons return to the ground state, they emit photons.
The figure below is the energy level diagram of atom X with three energy levels, E1 = 0.00eV, E2 = 1.78eV and E3 = 2.95eV. The ground state is considered 0 eV for reference. The transition of electrons takes place between levels E1 and E2.
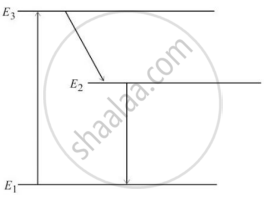
- What wavelength of radiation is needed to excite the atom to energy level E2 from E1?
- Suppose the external source has a power of 100 W. What would be the rate of photon emission?
