Advertisements
Advertisements
प्रश्न
Match the items of Column I and Column II.
| Column I | Column II | |
| (i) | SN1 reaction | (a) vic-dibromides |
| (ii) | Chemicals in fire extinguisher | (b) gem-dihalides |
| (iii) | Bromination of alkenes | (c) Racemisation |
| (iv) | Alkylidene halides | (d) Saytzeff rule |
| (v) | Elimination of HX from alkylhalide | (e) Chlorobromocarbons |
उत्तर
| Column I | Column II | |
| (i) | SN1 reaction | (c) Racemisation |
| (ii) | Chemicals in fire extinguisher | (e) Chlorobromocarbons |
| (iii) | Bromination of alkenes | (a) vic-dibromides |
| (iv) | Alkylidene halides | (b) gem-dihalides |
| (v) | Elimination of HX from alkylhalide | (d) Saytzeff rule |
Explanation:
(i) A mixture containing two enantiomers in equal proportions will have zero optical rotation, such a mixture is known as racemic mixture. The process of conversion of enantiomer into a racemic mixture is known as racemisation. If an alkyl halide follows SN1 mechanism then racemisation takes place while if it follows SN2 mechanism than inversion takes places.
(ii) Chlorobromocarbons are used in fire extinguishers.
(iii) In vicinal dihalides, halogen atoms are present on the adjacent carbon atom. Bromination of alkanes will give vicinal dihalides.
(iv) Alkylidene halides are named gem-dihalides. In gem-dihalides halogen atoms are present on same carbon atom.
(v) Elimination of HX from alkylhalide follows Saytzeff rule. This rule states that “in dehydrohalogenation reactions, the preferred product is that alkene which has the greater number of alkyl groups attached to the doubly bonded carbon atoms”.
APPEARS IN
संबंधित प्रश्न
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
m-ClCH2C6H4CH2C(CH3)3
When two halogen atoms are attached to the same carbon atom then it is:
m-Xylene reacts with Br2 in presence of FeBr3, what are products formed:
Which of the products will be major product in the reaction given below? Explain.
\[\ce{CH3CH = CH2 + HI -> \underset{(A)}{CH3CH2CH2I} + \underset{(B)}{CH3CHICH3}}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3CH2C(CH3)2CH2I}\]
Name the following halide according to IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(Cl)(C2H5)CH2CH3}\]
Name the following halide according to IUPAC system and classify as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides.
\[\ce{CH3CH2C(CH3)2CH2I}\]
Draw the structure of major monohalo products in the following reaction:
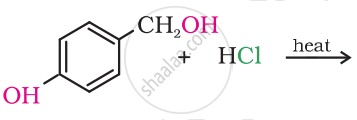
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
