Advertisements
Advertisements
प्रश्न
Mention the conditions required to maximise the yield of ammonia.
उत्तर १
Ammonia is prepared using the Haber’s process. The yield of ammonia can be maximized under the following conditions:
(i) High pressure (∼ 200 atm)
(ii) A temperature of ∼700 K
(iii) Use of a catalyst such as iron oxide mixed with small amounts of K2O and Al2O3
उत्तर २
Ammonia is prepared by Haber’s process as given below:
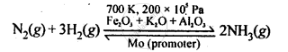
`triangle_rH^@ = -92.4 kJ mol^(-1)`
According to Le Chatelier's principle to maximise the yield of ammonia high P and T=~700 K should be used. The catalyst increases the rate of reaction and Mo promoter increase the efficiency of Fe catalyst.
APPEARS IN
संबंधित प्रश्न
Describe the laboratory method of preparation of ammonia
What is the action of Excess of air on ammonia ?
What is the action of the following reagents on ammonia :
Nessler's reagent
How does ammonia react with a solution of Cu2+?
What is the action of the following reagents on ammonia :
Sodium metal
What is Nessler’s reagent ?
On heating ammonium dichromate and barium azide separately we get ______.
The shape of ammonia molecule is ____________.
When ammonia is heated with cupric oxide, a molecule of ammonia will ____________.
Liquid ammonia bottles are opened after cooling them in ice for some time. It is because liquid NH3 ____________.
Ammonia is generally manufactured for fertilizers by the reaction:
Which one of the following is not a use of ammonia?
In qualitative analysis when \[\ce{H2S}\] is passed through an aqueous solution of salt acidified with dil. \[\ce{HCl}\], a black precipitate is obtained. On boiling the precipitate with dil. \[\ce{HNO3}\], it forms a solution of blue colour. Addition of excess of aqueous solution of ammonia to this solution gives ______.
A brown ring is formed in the ring test for \[\ce{NO3^{-}}\] ion. It is due to the formation of ______.
\[\ce{PCl5}\] reacts with finely divided silver on heating and a white silver salt is obtained, which dissolves on adding excess aqueous \[\ce{NH3}\] solution. Write the reactions involved to explain what happens.
What happens when reactions:
Benzylchloride is treated with ammonia followed by the reaction with Chloromethane.
