Advertisements
Advertisements
प्रश्न
Mention the two features of x-ray spectra, not explained by classical electromagnetic theory.
उत्तर
Though classical electromagnetic theory suggests the emission of radiations from accelerating electrons, it could not explain two features exhibited by x-ray spectra. These features are given below
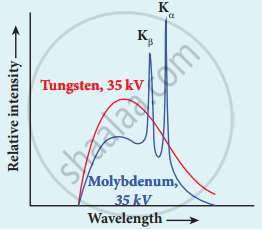
- For a given accelerating voltage, the lower limit for the wavelength of continuous x-ray spectra is the same for all targets. This minimum wavelength is called cut-off wavelength.
- The intensity of x-rays is significantly increased at certain well-defined wavelengths as shown in the case of characteristic x-ray spectra for molybdenum.
APPEARS IN
संबंधित प्रश्न
If the mean wavelength of light from sun is taken as 550 nm and its mean power as 3.8 × 1026 W, then the average number of photons received by the human eye per second from sunlight is of the order of
A light of wavelength 500 nm is incident on a sensitive metal plate of photoelectric work function 1.235 eV. The kinetic energy of the photoelectrons emitted is (Take h = 6.6 × 10–34 Js)
Give the definition of intensity of light according to quantum concept and its unit.
Explain how frequency of incident light varies with stopping potential.
List out the laws of photoelectric effect.
Obtain Einstein’s photoelectric equation with the necessary explanation.
List out the characteristics of photons.
A 3310 Å photon liberates an electron from a material with energy 3 × 10−19 J while another 5000 Å photon ejects an electron with energy 0.972 × 10−19 J from the same material. Determine the value of Planck’s constant and the threshold wavelength of the material.
At the given point of time, the earth receives energy from the sun at 4 cal cm–2 min–1. Determine the number of photons received on the surface of the Earth per cm2 per minute. (Given: Mean wavelength of sunlight = 5500 Å)
UV light of wavelength 1800 Å is incident on a lithium surface whose threshold wavelength is 4965 Å. Determine the maximum energy of the electron emitted.
