Advertisements
Advertisements
प्रश्न
Name the substance which on treatment with chlorine yields bleaching powder?
उत्तर
Calcium hydroxide [Ca(OH)2]
APPEARS IN
संबंधित प्रश्न
When you add sodium hydrogen carbonate to acetic acid in a test tube, a gas liberates immediately with brisk effervescence. Name this gas. Describe the method of testing this gas.
What will happen if a solution of sodium hydrocarbonate is heated? Give the equation of the reaction involved.
Name an acid which is present in baking powder.
Complete and balance the following chemical equations:
`NaHCO_3` 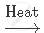
What is baking soda?
Write the chemical name of baking soda. Give the important uses of baking soda. How does baking soda differ chemically from washing soda?
What is the common name of sodium hydrogencarbonate?
How does baking powder differ from baking soda?
What are the materials used for the preparation of bleaching powder?
In addition to sodium hydrogencarbonate, baking powders contain a substance X. Name the substance X. What is the role of substance X in the baking powder?
The formula of baking soda is:
(a) K2CO3
(b) KHCO3
(c) NaHCO3
(d) Na2CO3
Consider the following substances:
NaCl, Ca(OH)2, NaHCO3, NH3, Na2CO3, H2O, Cl2, CO2, CaSO4.2H2O, 2CaSO4.H2O, CaOCl2
Which compound is a part of baking powder?
(i) Name the products formed when sodium hydrogen carbonate is heated.
(ii) Write the chemical equation for the reaction involved in the above.
Baking soda is a mixture of:
Salt 'P', commonly used in bakery products, on heating gets converted into another salt 'Q' which itself is used for the removal of hardness of water and a gas 'R' is evolved. The gas 'R', when passed through freshly prepared lime water, turns milky. Identify 'P', 'Q' and 'R', giving a chemical equation for the justification of your answer.
Mild non-corrosive basic salt is ______.
