Advertisements
Advertisements
प्रश्न
Name the following compound according to IUPAC system.
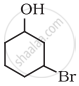
उत्तर
3-Bromocyclohexanol
APPEARS IN
संबंधित प्रश्न
Explain metamerism with suitable examples of ethers
Name the following compound according to IUPAC system.
\[\begin{array}{cc}
\ce{H2C = CH - CH - CH2 - CH2 - CH3}\\
|\phantom{..........}\\
\ce{OH}\phantom{........}
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{.}}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{H3C - CH - CH2 - CH - CH - CH2 - CH3}\\
\phantom{}|\phantom{.............}|\phantom{......}|\phantom{.........}\\
\phantom{}\ce{OH}\phantom{..........}\ce{OH}\phantom{...}\ce{C2H5}\phantom{......}
\end{array}\]
Write IUPAC name of the following compound:
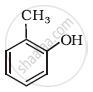
Write structures of the compounds whose IUPAC names are as follows:
3-Chloromethylpentan-1-ol.
Give IUPAC name of the following ether:
CH3CH2CH2OCH3
Write IUPAC name of the following compound (CH3)2 N − CH2CH3
Write the structures of the products when Butan-2-ol reacts with CrO3
What.will be the product fonned when chlorobenzene is heated with sodium metal in the presence of dry ether?
In a carbinol system of nomenclature tert.butyl alcohol is named as _______________
One of the following is not a dihydroxy derivative of benzene.
An example of a compound with functional group – O – is ____________.
Which of the following compounds is oxidised to prepare methyl ethyl ketone?
Ethylene reacts with Baeyer’s reagent to give ______.
\[\ce{Phenol ->[Zn, dust] 'X' ->[CH3Cl][Anhy. AlCl3] 'Y' ->[Alkaline][KMnO4] 'Z'}\]
The product ‘Z’ is:
IUPAC name of the compound is:
\[\begin{array}{cc}
\ce{CH3-CH-OCH3}\\
|\phantom{....}\\
\ce{CH3}\phantom{..}
\end{array}\]
Arrange the following compounds in decreasing order of acidity.
\[\ce{H2O, ROH, HC ≡ CH}\]
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Draw structure of the following compound.
2. 5-Diethylphenol
