Advertisements
Advertisements
प्रश्न
Arrange the following compounds in decreasing order of acidity.
\[\ce{H2O, ROH, HC ≡ CH}\]
उत्तर
The decreasing order of the acidity of the given compounds:
\[\ce{H2O > ROH > HC ≡ CH}\]
\[\ce{HC ≡ CH}\] is less acidic because the carbon atoms here is sp hybridized, so the electron density is higher on the carbon atom.
APPEARS IN
संबंधित प्रश्न
Give IUPAC name of the following ether:
CH3OCH2CH2Cl
How is phenol converted into the following?
benzene
Write the structures of the products when Butan-2-ol reacts with SOCl2
Write the IUPAC name of the following :
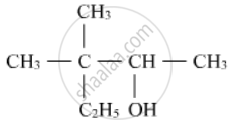
Write structural formulae for Methyl vinyl ether.
In a carbinol system of nomenclature tert.butyl alcohol is named as _______________
Assertion: Phenols give o- and p-nitrophenol on nitration with conc. \[\ce{HNO3}\] and \[\ce{H2SO4}\] mixture.
Reason: –OH group in phenol is o–, p– directing.
Draw structure of the following compound.
2. 5-Diethylphenol
The IUPAC name of 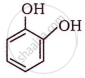 is ______.
is ______.
Write IUPAC names of the following compounds:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{...}OH\phantom{...}CH3}\\
\end{array}\]
