Advertisements
Advertisements
Question
Name the following compound according to IUPAC system.
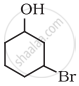
Solution
3-Bromocyclohexanol
APPEARS IN
RELATED QUESTIONS
Write the IUPAC name of the given compound:
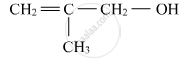
Name the following compound according to IUPAC system.
\[\begin{array}{cc}
\ce{CH3 - C = C - CH2OH}\\
\phantom{}|\phantom{....}|\phantom{....}\\
\phantom{}\ce{CH3}\phantom{.}\ce{Br}\phantom{...}
\end{array}\]
Write structures of the compounds whose IUPAC names are as follows:
3-Chloromethylpentan-1-ol.
- Draw the structures of all isomeric alcohols of molecular formula C5H12O and give their IUPAC names.
- Classify the isomers of alcohols in the above question as primary, secondary and tertiary alcohols.
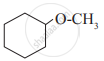
Give IUPAC name of the following ether:
O2N – C6H4 – OCH3(p)
Natalite is a mixture of
(a) diethyl ether and methanol
(b) diethyl ether and ethanol
(c) dimethyl ether and methanol
(d) dimethyl ether and ethanol
Write the IUPAC name of the following compound:
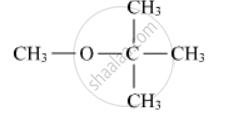
What.will be the product fonned when chlorobenzene is heated with sodium metal in the presence of dry ether?
Write IUPAC names of the following
In a carbinol system of nomenclature tert.butyl alcohol is named as _______________
Cresol has ____________.
The product of acid catalysed hydration of 2-phenylpropene is:
The heating of phenyl methyl ether with HI produces:
Which of the following compounds will react with sodium hydroxide solution in water?
Assertion: Phenol forms 2, 4, 6 – tribromophenol on treatment with \[\ce{Br2}\] in carbon disulphide at 273 K.
Reason: Bromine polarises in carbon disulphide.
Explain why Lewis acid is not required in bromination of phenol?
Write structural formulae for:
Salicylic acid
Write IUPAC names of the following compounds:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{...}OH\phantom{...}CH3}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
