Advertisements
Advertisements
Question
Which of the following compounds will react with sodium hydroxide solution in water?
Options
\[\ce{C6H5OH}\]
\[\ce{C6H5CH2OH}\]
\[\ce{(CH3)3COH}\]
\[\ce{C2H5OH}\]
Solution
\[\ce{C6H5OH}\]
Explanation:
Phenol being more acidic reacts with sodium hydroxide solution in water to give sodium phenoxide which is resonance stabilized.
Alcohols are very weak acids.
\[\ce{C6H5OH + NaOH -> C6H5ONa + H2O}\]
APPEARS IN
RELATED QUESTIONS
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH3}\\
|\phantom{......}|\phantom{..}\\
\ce{OH}\phantom{...}\ce{OH}\phantom{}
\end{array}\]
Write IUPAC name of the following compound (CH3)2 N − CH2CH3
Write the IUPAC name of the following compound:
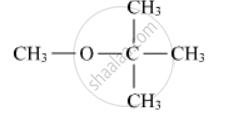
Write the structures of the products when Butan-2-ol reacts with CrO3
Write structural formulae for Methyl vinyl ether.
In a carbinol system of nomenclature tert.butyl alcohol is named as _______________
Give IUPAC names of the following compound:
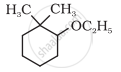
\[\ce{HC ≡ CH ->[HgSO4][H2SO4] ->[CH3MgBr][H2O] ->[PBr3]}\]
Write chemical reactions for the following conversion:
Acetic acid into ethyl alcohol
Write IUPAC names of the following compounds:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{...}OH\phantom{...}CH3}\\
\end{array}\]
