Advertisements
Advertisements
प्रश्न
Observe the graph shown in figure and answer the following questions:
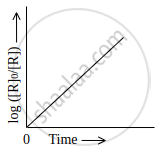
- What is the order of the reaction?
- What is the slope of the curve?
- Write the relationship between k and t1/2 (half life period).
उत्तर
- The order of the reaction is first-order reaction.
- Slope of the curve = `k/2.303`
- The half-life for the first-order reaction is given by t1/2 = `0.693/k`
APPEARS IN
संबंधित प्रश्न
Sucrose decomposes in acid solution into glucose and fructose according to the first order rate law with `"t"_(1/2)`= 3 hours. What fraction of the sample of sucrose remains after 8 hours?
The following data were obtained during the first order thermal decomposition of SO2Cl2 at a constant volume.
\[\ce{SO2Cl2_{(g)} -> SO2_{(g)} + Cl2_{(g)}}\]
| Experiment | Time/s–1 | Total pressure/atm |
| 1 | 0 | 0.5 |
| 2 | 100 | 0.6 |
Calculate the rate of the reaction when total pressure is 0.65 atm.
For the decomposition of azoisopropane to hexane and nitrogen at 543 K, the following data are obtained.
| t (sec) | P(mm of Hg) |
| 0 | 35.0 |
| 360 | 54.0 |
| 720 | 63.0 |
Calculate the rate constant.
Which of the following graphs is correct for a first order reaction?

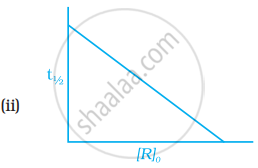
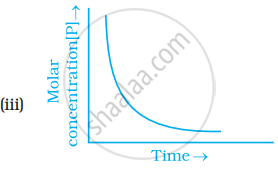
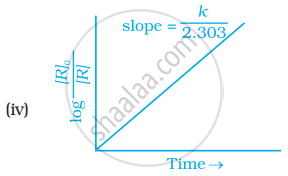
First order reaction is 50% complete in 1.26 × 1014s. How much time could it take for 100% completion?
Gaseous cyclobutene isomerizes to butadiene in a first order process which has a 'k' value of 3.3 × 10−4 s−1 at 153°C. The time in minutes it takes for the isomerization to proceed 40% to completion at this temperature is ______. (Rounded-off to the nearest integer)
How will you represent first order reactions graphically?
The rate constant for the reaction:
\[\ce{2N2O_{(s)} ->2N2O4_{(g)}}\] is 4.98 × 10-4 s-1.
The order of the reaction is ______.
Write the equation for integrated rate law for a first order reaction.
Show that `t_(1/2)= 0.693/k` for first reaction.
