Advertisements
Advertisements
प्रश्न
238U decays to 206Pb with a half-life of 4.47 × 109 y. This happens in a number of steps. Can you justify a single half for this chain of processes? A sample of rock is found to contain 2.00 mg of 238U and 0.600 mg of 206Pb. Assuming that all the lead has come from uranium, find the life of the rock.
उत्तर
Given:
Half-life of 238U, t1/2 = 4.47 × 109 years
Total Number of atoms present in the rock initially , `N_0 = ((6.023 xx 10^23 xx 2)/238 + (6.023 xx 10^23 xx 0.6)/206)`
= `(12.046/238 + 3.62/206) xx 10^20`
= `(0.0505 + 0.0175) xx 10^20`
= `0.0680 xx 10^20`
Now , `N = N_0e^(-lambdat)`
Here, `lambda` = Disintegration constant
t = Life of the rock
⇒ `N = N_0e^((-0.693)/(t/"1/2") xx t)`
⇒ `0.0505 = 0.0680e^((-0.693)/(4.47 xx 10^9) xx t)`
⇒ `"In" (0.0505/0.0680) = (-06931)/(4.47 xx 10^9) xx t`
⇒ `t = 1.92 xx 10^9` years
APPEARS IN
संबंधित प्रश्न
The half life of a certain radioactive material against \u0003α-decay is 100 days. After how much time, will the undecayed fraction of the material be 6.25%?
Write nuclear reaction equation for β−-decay of `""_83^210"Bi"`.
Write nuclear reaction equation for β+-decay of `""_6^11"C"`.
Write nuclear reaction equation for electron capture of `""_54^120"Xe"`.
A radioactive nucleus has a decay constant λ = 0.3465 (day)–1. How long would it take the nucleus to decay to 75% of its initial amount?
The sequence of stepwise decay of a radioactive nucleus is
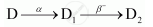
If the atomic number and mass number of D2 are 71 and 176 respectively, what are their corresponding values of D?
The half-life of 199Au is 2.7 days. (a) Find the activity of a sample containing 1.00 µg of 198Au. (b) What will be the activity after 7 days? Take the atomic weight of 198Au to be 198 g mol−1.
Radioactive 131I has a half-life of 8.0 days. A sample containing 131I has activity 20 µCi at t = 0. (a) What is its activity at t = 4 days? (b) What is its decay constant at t = 4.0 days?
A certain sample of a radioactive material decays at the rate of 500 per second at a certain time. The count rate falls to 200 per second after 50 minutes. (a) What is the decay constant of the sample? (b) What is its half-life?
The half-life of 226Ra is 1602 y. Calculate the activity of 0.1 g of RaCl2 in which all the radium is in the form of 226Ra. Taken atomic weight of Ra to be 226 g mol−1 and that of Cl to be 35.5 g mol−1.
The half-life of a radioisotope is 10 h. Find the total number of disintegration in the tenth hour measured from a time when the activity was 1 Ci.
Natural water contains a small amount of tritium (`""_1^3H`). This isotope beta-decays with a half-life of 12.5 years. A mountaineer while climbing towards a difficult peak finds debris of some earlier unsuccessful attempt. Among other things he finds a sealed bottled of whisky. On returning, he analyses the whisky and finds that it contains only 1.5 per cent of the `""_1^3H` radioactivity as compared to a recently purchased bottle marked '8 years old'. Estimate the time of that unsuccessful attempt.
A human body excretes (removes by waste discharge, sweating, etc.) certain materials by a law similar to radioactivity. If technetium is injected in some form in a human body, the body excretes half the amount in 24 hours. A patient is given an injection containing 99Tc. This isotope is radioactive with a half-life of 6 hours. The activity from the body just after the injection is 6 μCi. How much time will elapse before the activity falls to 3 μCi?
`""_83^212"Bi"` can disintegrate either by emitting an α-particle of by emitting a β−-particle. (a) Write the two equations showing the products of the decays. (b) The probabilities of disintegration α-and β-decays are in the ratio 7/13. The overall half-life of 212Bi is one hour. If 1 g of pure 212Bi is taken at 12.00 noon, what will be the composition of this sample at 1 P.m. the same day?
In a gamma ray emission from nucleus :
The half-life of a certain radioactive element is 3.465 days. Find its disintegration constant.
A nucleus with Z = 92 emits the following in a sequence:
α, β‾, β‾, α, α, α, α, α, β‾, β‾, α, β+, β+, α
Then Z of the resulting nucleus is ______.
