Advertisements
Advertisements
प्रश्न
Natural water contains a small amount of tritium (`""_1^3H`). This isotope beta-decays with a half-life of 12.5 years. A mountaineer while climbing towards a difficult peak finds debris of some earlier unsuccessful attempt. Among other things he finds a sealed bottled of whisky. On returning, he analyses the whisky and finds that it contains only 1.5 per cent of the `""_1^3H` radioactivity as compared to a recently purchased bottle marked '8 years old'. Estimate the time of that unsuccessful attempt.
उत्तर
Given:
Half-life time of tritium, `T_"1/2"` = 12.5 years
Disintegration constant, `lambda = 0.693/12.5` per year
Let A0 be the activity, when the bottle was manufactured.
Activity after 8 years (A) is given by
`A = A_0e^((-0.693)/(12.5) xx 8)` .....(1)
Let us consider that the mountaineering had taken place t years ago.
Then, activity of the bottle (A') on the mountain is given by
`A' = A_0e^(-lambdat)`
Here, A' = (Activity of the bottle manufactured 8 years ago) × 1.5 %
`A' = A_0e^((-0.693)/(12.5) xx 8) xx 0.015` ...(2)
Comparing (1) and (2)
`(-0.693)/12.5 t = (-0.6931 xx 8)/12.5 + "In" [0.015]`
⇒ `(-0.693)/12.5 t = (-0.693)/12.5 xx 8 - 4.1997`
⇒ `0.639 t = 58.040`
⇒ t = 83.75 years
APPEARS IN
संबंधित प्रश्न
The half life of a certain radioactive material against \u0003α-decay is 100 days. After how much time, will the undecayed fraction of the material be 6.25%?
Write nuclear reaction equation for α-decay of `""_88^226"Ra"`.
Write nuclear reaction equation for α-decay of `""_94^242"Pu"`.
Write nuclear reaction equation for β−-decay of `""_83^210"Bi"`.
Write nuclear reaction equation for β+-decay of `""_43^97"Tc"`.
Write nuclear reaction equation for electron capture of `""_54^120"Xe"`.
Draw graphs showing variation of photoelectric current with applied voltage for two incident radiations of equal frequency and different intensities. Mark the graph for the radiation of higher intensity.
A radioactive nucleus has a decay constant λ = 0.3465 (day)–1. How long would it take the nucleus to decay to 75% of its initial amount?
The sequence of stepwise decay of a radioactive nucleus is
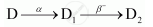
If the atomic number and mass number of D2 are 71 and 176 respectively, what are their corresponding values of D?
State the law of radioactive decay. hence derive the relation N = Noe-λt . Represent it graphically.
The half-life of 199Au is 2.7 days. (a) Find the activity of a sample containing 1.00 µg of 198Au. (b) What will be the activity after 7 days? Take the atomic weight of 198Au to be 198 g mol−1.
Radioactive 131I has a half-life of 8.0 days. A sample containing 131I has activity 20 µCi at t = 0. (a) What is its activity at t = 4 days? (b) What is its decay constant at t = 4.0 days?
The half-life of a radioisotope is 10 h. Find the total number of disintegration in the tenth hour measured from a time when the activity was 1 Ci.
Radioactive isotopes are produced in a nuclear physics experiment at a constant rate dN/dt = R. An inductor of inductance 100 mH, a resistor of resistance 100 Ω and a battery are connected to form a series circuit. The circuit is switched on at the instant the production of radioactive isotope starts. It is found that i/N remains constant in time where i is the current in the circuit at time t and N is the number of active nuclei at time t. Find the half-life of the isotope.
In a gamma ray emission from nucleus :
Copy and complete the following table for a radioactive element whose half-life is 10 minutes. Assume that you have 30g of this element at t = 0.

Plot a graph showing the variation of undecayed nuclei N versus time t. From the graph, find out how one can determine the half-life and average life of the radioactive nuclei.
Half-life of a certain radioactive material is 8 hours.
Find the disintegration constant of this material.
