Advertisements
Advertisements
प्रश्न
The sequence of stepwise decay of a radioactive nucleus is
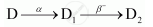
If the atomic number and mass number of D2 are 71 and 176 respectively, what are their corresponding values of D?
उत्तर

So, the corresponding values of atomic number and mass number for D are 74 and 180.
APPEARS IN
संबंधित प्रश्न
For the past some time, Aarti had been observing some erratic body movement, unsteadiness and lack of coordination in the activities of her sister Radha, who also used to complain of severe headache occasionally. Aarti suggested to her parents to get a medical check-up of Radha. The doctor thoroughly examined Radha and diagnosed that she has a brain tumour.
(a) What, according to you, are the values displayed by Aarti?
(b) How can radioisotopes help a doctor to diagnose brain tumour?
Write nuclear reaction equation for α-decay of `""_88^226"Ra"`.
Write nuclear reaction equation for β−-decay of `""_15^32"P"`.
Write nuclear reaction equation for β+-decay of `""_43^97"Tc"`.
Radioactive 131I has a half-life of 8.0 days. A sample containing 131I has activity 20 µCi at t = 0. (a) What is its activity at t = 4 days? (b) What is its decay constant at t = 4.0 days?
A certain sample of a radioactive material decays at the rate of 500 per second at a certain time. The count rate falls to 200 per second after 50 minutes. (a) What is the decay constant of the sample? (b) What is its half-life?
The half-life of a radioisotope is 10 h. Find the total number of disintegration in the tenth hour measured from a time when the activity was 1 Ci.
Natural water contains a small amount of tritium (`""_1^3H`). This isotope beta-decays with a half-life of 12.5 years. A mountaineer while climbing towards a difficult peak finds debris of some earlier unsuccessful attempt. Among other things he finds a sealed bottled of whisky. On returning, he analyses the whisky and finds that it contains only 1.5 per cent of the `""_1^3H` radioactivity as compared to a recently purchased bottle marked '8 years old'. Estimate the time of that unsuccessful attempt.
A sample contains a mixture of 108Ag and 110Ag isotopes each having an activity of 8.0 × 108 disintegration per second. 110Ag is known to have larger half-life than 108Ag. The activity A is measured as a function of time and the following data are obtained.
| Time (s) |
Activity (A) (108 disinte- grations s−1) |
Time (s) |
Activity (A 108 disinte-grations s−1) |
| 20 40 60 80 100 |
11.799 9.1680 7.4492 6.2684 5.4115 |
200 300 400 500 |
3.0828 1.8899 1.1671 0.7212 |
(a) Plot ln (A/A0) versus time. (b) See that for large values of time, the plot is nearly linear. Deduce the half-life of 110Ag from this portion of the plot. (c) Use the half-life of 110Ag to calculate the activity corresponding to 108Ag in the first 50 s. (d) Plot In (A/A0) versus time for 108Ag for the first 50 s. (e) Find the half-life of 108Ag.
The half-life of a certain radioactive element is 3.465 days. Find its disintegration constant.
