Advertisements
Advertisements
प्रश्न
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroxylation using bayers reagent
उत्तर
\[\begin{array}{cc}
\phantom{.........................................}\ce{OH}\phantom{.}\ce{OH}\phantom{..}\\
\phantom{......................................}|\phantom{....}|\phantom{}\\
\ce{CH3 - C = CH - CH3 ->[Cold KMnO4/OH^Θ][(O)] 3CH3 - C - CH - CH3}\\
\phantom{..}|\phantom{.......................................}|\phantom{........}\\
\phantom{.}\ce{\underset{(2-methyl-but-2-ene)}{CH3}}\phantom{......................}\ce{\underset{(2-methyl-butan-2, 3-diol)}{CH3}}\phantom{..}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
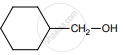 on treatment with Con. H2SO4 predominately gives
on treatment with Con. H2SO4 predominately gives
Carbolic acid is ____________.
Which of the following compound can be used as antifreeze in automobile radiators?
One mole of an organic compound (A) with the formula C3H8O reacts completely with two moles of HI to form X and Y. When Y is boiled with aqueous alkali it forms Z. Z answers the iodoform test. The compound (A) is ___________.
Write the structure of the aldehyde, carboxylic acid and ester that yield 4-methylpent-2-en-1-ol.
0.44 g of a monohydric alcohol, when added to methyl magnesium iodide in ether, liberates at STP 112 cm3 of methane with PCC the same alcohol form a carbonyl compound that answers the silver mirror test. Identify the compound.
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI
Identify the product (s) is / are formed when 1 – methoxy propane is heated with excess HI. Name the mechanism involved in the reaction
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
The correct IUPAC name of the compound,
\[\begin{array}{cc}
\ce{CH3}\phantom{......}\\
|\phantom{........}\\
\ce{H3C - CH - CH - CH - CH2 - OH}\\
|\phantom{............}|\phantom{........}\\
\ce{Cl}\phantom{...........}\ce{CH3}\phantom{......}
\end{array}\]
