Advertisements
Advertisements
Question
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroxylation using bayers reagent
Solution
\[\begin{array}{cc}
\phantom{.........................................}\ce{OH}\phantom{.}\ce{OH}\phantom{..}\\
\phantom{......................................}|\phantom{....}|\phantom{}\\
\ce{CH3 - C = CH - CH3 ->[Cold KMnO4/OH^Θ][(O)] 3CH3 - C - CH - CH3}\\
\phantom{..}|\phantom{.......................................}|\phantom{........}\\
\phantom{.}\ce{\underset{(2-methyl-but-2-ene)}{CH3}}\phantom{......................}\ce{\underset{(2-methyl-butan-2, 3-diol)}{CH3}}\phantom{..}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Isopropyl benzene on air oxidation in the presence of dilute acid gives ___________.
Which of the following compound can be used as antifreeze in automobile radiators?
One mole of an organic compound (A) with the formula C3H8O reacts completely with two moles of HI to form X and Y. When Y is boiled with aqueous alkali it forms Z. Z answers the iodoform test. The compound (A) is ___________.
How is the conversion effected benzyl alcohol to benzoic acid?
0.44 g of a monohydric alcohol, when added to methyl magnesium iodide in ether, liberates at STP 112 cm3 of methane with PCC the same alcohol form a carbonyl compound that answers the silver mirror test. Identify the compound.
Complete the following reaction.
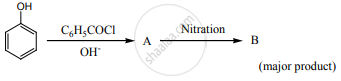
Phenol is distilled with Zn dust followed by Friedel-crafts alkylation with propyl chloride to give a compound B, B on oxidation gives (c) Identify A, B and C.
\[\begin{array}{cc}\phantom{..........}\ce{CH3}\\\phantom{.......}/\\\ce{C6H4}\\
\phantom{.......}\backslash\\\phantom{.........}\ce{OH}\end{array}\] is a/an ______.
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI
What will be the product (X and A) for the following reaction.
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O^+] X ->[acid K2Cr2O7] A}\]
