Advertisements
Advertisements
Question
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroboration
Solution
\[\begin{array}{cc}
\phantom{...............................................}\ce{CH3}\\
\phantom{............................................}|\\
\ce{6CH3 - C = CH - CH3 + B2H6 -> 2[(CH3)2 CH - CH -]_3 B}\\
|\phantom{..........................................}\\
\ce{\underset{(2-methyl-but-2-ene)}{CH3}}\phantom{.......................................}
\end{array}\]
\[\begin{array}{cc}
\phantom{..}\ce{CH3}\phantom{............................}\ce{CH3}\phantom{...}\ce{CH3}\phantom{...}\\
\phantom{.}|\phantom{................................}|\phantom{......}|\phantom{....}\\
\ce{[(CH3)2 CH - CH -]_3 B + 3H2O2 ->[OH^Θ] \underset{(3-methyl-butan-2-ol)}{3CH3 - CH - CH - OH} + H3BO3}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Which one of the following will react with phenol to give salicylaldehyde after hydrolysis.
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroxylation using bayers reagent
Is it possible to oxidise t-butyl alcohol using acidified dichromate to form a carbonyl compound?
Write the chemical equation for Williamson's synthesis of 2-ethoxy-2-methyl pentane starting from ethanol and 2-methyl pentan-2-ol.
How is the conversion effected benzyl chloride to benzyl alcohol?
How is the conversion effected benzyl alcohol to benzoic acid?
0.44 g of a monohydric alcohol, when added to methyl magnesium iodide in ether, liberates at STP 112 cm3 of methane with PCC the same alcohol form a carbonyl compound that answers the silver mirror test. Identify the compound.
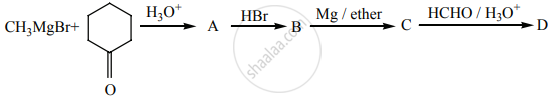
Identify A, B, C, D and write the complete equation.
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in each of the following method.
Acid catalysed hydration
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
