Advertisements
Advertisements
प्रश्न
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroboration
उत्तर
\[\begin{array}{cc}
\phantom{...............................................}\ce{CH3}\\
\phantom{............................................}|\\
\ce{6CH3 - C = CH - CH3 + B2H6 -> 2[(CH3)2 CH - CH -]_3 B}\\
|\phantom{..........................................}\\
\ce{\underset{(2-methyl-but-2-ene)}{CH3}}\phantom{.......................................}
\end{array}\]
\[\begin{array}{cc}
\phantom{..}\ce{CH3}\phantom{............................}\ce{CH3}\phantom{...}\ce{CH3}\phantom{...}\\
\phantom{.}|\phantom{................................}|\phantom{......}|\phantom{....}\\
\ce{[(CH3)2 CH - CH -]_3 B + 3H2O2 ->[OH^Θ] \underset{(3-methyl-butan-2-ol)}{3CH3 - CH - CH - OH} + H3BO3}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Which of the following compounds on reaction with methyl magnesium bromide will give tertiary alcohol.
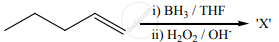
The X is
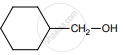 on treatment with Con. H2SO4 predominately gives
on treatment with Con. H2SO4 predominately gives
Carbolic acid is ____________.
Isopropyl benzene on air oxidation in the presence of dilute acid gives ___________.
Arrange the following in the increasing order of their boiling point and give a reason for your ordering.
Propan-1-ol, propan-1, 2, 3-triol, propan-1, 3-diol, propan-2-ol
What will be the product (X and A) for the following reaction?
acetylchloride \[\ce{->[i) CH3MgBr][ii) H3O+] X ->[acid K2Cr2O7] A}\]
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
What will be the product (X and A) for the following reaction.
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O^+] X ->[acid K2Cr2O7] A}\]
The correct IUPAC name of the compound,
\[\begin{array}{cc}
\ce{CH3}\phantom{......}\\
|\phantom{........}\\
\ce{H3C - CH - CH - CH - CH2 - OH}\\
|\phantom{............}|\phantom{........}\\
\ce{Cl}\phantom{...........}\ce{CH3}\phantom{......}
\end{array}\]
