Advertisements
Online Mock Tests
Chapters
2: p-Block Elements - I
3: p-Block Elements - II
4: Transition and Inner Transition Elements
5: Coordination Chemistry
6: Solid State
7: Chemical Kinetics
8: Ionic Equilibrium
9: Electro Chemistry
10: Surface Chemistry
▶ 11: Hydroxy Compounds and Ethers
12: Carbonyl Compounds and Carboxylic Acids
13: Organic Nitrogen Compounds
14: Biomolecules
15: Chemistry in Everyday Life
![Samacheer Kalvi solutions for Chemistry - Volume 1 and 2 [English] Class 12 TN Board chapter 11 - Hydroxy Compounds and Ethers Samacheer Kalvi solutions for Chemistry - Volume 1 and 2 [English] Class 12 TN Board chapter 11 - Hydroxy Compounds and Ethers - Shaalaa.com](/images/chemistry-volume-1-and-2-english-class-12-tn-board_6:5f2b1b2038084cf381bfa42c826a928c.jpg)
Advertisements
Solutions for Chapter 11: Hydroxy Compounds and Ethers
Below listed, you can find solutions for Chapter 11 of Tamil Nadu Board of Secondary Education Samacheer Kalvi for Chemistry - Volume 1 and 2 [English] Class 12 TN Board.
Samacheer Kalvi solutions for Chemistry - Volume 1 and 2 [English] Class 12 TN Board 11 Hydroxy Compounds and Ethers Evaluation [Pages 139 - 143]
Choose the correct answer:
An alcohol (x) gives blue colour in Victormeyer’s test and 3.7 g of X when treated with metallic sodium liberates 560 mL of hydrogen at 273 K and 1 atm pressure what will be the possible structure of X?
CH3CH(OH)CH2CH3
CH3 – CH(OH) – CH3
CH3 – C(OH) (CH3)2
CH3 – CH2 – CH(OH) – CH2 – CH3
Which of the following compounds on reaction with methyl magnesium bromide will give tertiary alcohol.
benzaldehyde
propanoic acid
methyl propanoate
acetaldehyde
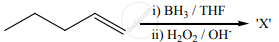
The X is
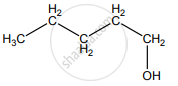
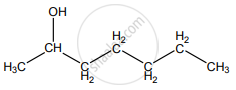
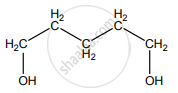
None of these
In the reaction sequence, \[\ce{Ethene ->[HOCl] A ->[X] ethan-1, 2-diol}\]. A and X respectively are ____________.
Chloroethane and NaOH
ethanol and H2SO4
2-chloroethane -1-ol and NaHCO3
ethanol and H2O
Which one of the following is the strongest acid.
2-nitrophenol
4-chlorophenol
4-nitrophenol
3-nitrophenol
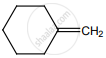
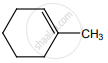
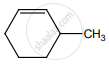
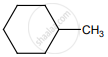
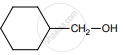 on treatment with Con. H2SO4 predominately gives
on treatment with Con. H2SO4 predominately gives
Carbolic acid is ____________.
Phenol
Picric acid
benzoic acid
phenylacetic acid
Which one of the following will react with phenol to give salicylaldehyde after hydrolysis.
Dichloro methane
trichloroethane
trichloro methane
CO2
\[\ce{(CH3)3 - C - CH(OH) CH3 ->[con H2SO4] X (major product)}\]
(CH3)3 CCH = CH2
(CH3)2 C = C (CH3)2
CH2 = C(CH3)CH2 – CH2 – CH3
CH2 = C (CH3) – CH2 – CH2 – CH3
The correct IUPAC name of the compound,
\[\begin{array}{cc}
\ce{CH3}\phantom{.....}\\
|\phantom{........}\\
\ce{H3C - CH - CH - CH - CH2 - OH}\\
\phantom{}|\phantom{............}|\phantom{........}\\
\phantom{}\ce{Cl}\phantom{..........}\ce{CH3}\phantom{......}
\end{array}\]
4-chloro-2, 3-dimethyl pentan-1-ol
2, 3-dimethyl-4-chloropentan-1-ol
2, 3, 4-trimethyl-4-chlorobutan-1-ol
4-chloro-2, 3, 4-trimethyl pentan-1-ol
Assertion: Phenol is more acidic than ethanol.
Reason: Phenoxide ion is resonance stabilized.
both assertion and reason are true and reason is the correct explanation of assertion.
both assertion and reason are true but reason is not the correct explanation of assertion.
assertion is true but reason is false.
both assertion and reason are false.
In the reaction \[\ce{Ethanol ->[PCl5] X ->[alc. KOH] Y ->[H2SO4/H2O][298 K] Z}\]. The ‘Z’ is
ethane
ethoxyethane
ethylbisulphite
ethanol
The reaction can be classified as
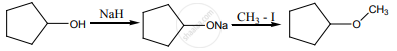
dehydration
Williamson alcohol synthesis
Williamson ether synthesis
dehydrogenation of alcohol
Isopropyl benzene on air oxidation in the presence of dilute acid gives ___________.
C6H5COOH
C6H5COCH3
C6H5COC6H5
C6H5 – OH
Assertion: Phenol is more reactive than benzene towards electrophilic substitution reaction
Reason: In the case of phenol, the intermediate arenium ion is more stabilized by resonance.
if both assertion and reason are true and reason is the correct explanation of assertion.
if both assertion and reason are true but reason is not the correct explanation of assertion.
assertion is true but reason is false.
both assertion and reason are false.
HO CH2 CH2 – OH on heating with periodic acid gives ____________.
methanoic acid
Glyoxal
methanal
CO2
Which of the following compound can be used as antifreeze in automobile radiators?
methanol
ethanol
Neopentyl alcohol
ethan-1, 2-diol
The reaction is an example of

Wurtz reaction
cyclic reaction
Williamson reaction
Kolbe reaction
One mole of an organic compound (A) with the formula C3H8O reacts completely with two moles of HI to form X and Y. When Y is boiled with aqueous alkali it forms Z. Z answers the iodoform test. The compound (A) is ___________.
propan-2-ol
propan-1-ol
ethoxy ethane
methoxy ethane
Among the following ethers which one will produce methyl alcohol on treatment with hot HI?
\[\ce{(H3C)3 C - O - CH3}\]
\[\ce{(CH3)2 - CH - CH2 - O - CH3}\]
\[\ce{CH3 (CH2)3 - O - CH3}\]
\[\begin{array}{cc}
\ce{CH3 - CH2 - CH - O - CH3}\\
|\\
\phantom{...}\ce{CH3}
\end{array}\]
Williamson's synthesis of preparing dimethyl ether is a/an ____________.
SN1 reactions
SN2 reaction
electrophilic addition
electrophilic substitution
On reacting with neutral ferric chloride, phenol gives ____________.
red colour
violet colour
dark green colour
no colouration
Short Answer Questions:
Identify the product(s) is/are formed when 1-methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
Suggest a suitable reagent to prepare secondary alcohol with the identical group using a Grignard reagent.
What is the major product obtained when two moles of ethyl magnesium bromide is treated with methyl benzoate followed by acid hydrolysis.
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Acid catalysed hydration
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroboration
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroxylation using bayers reagent
Arrange the following in the increasing order of their boiling point and give a reason for your ordering.
Butan-2-ol, Butan-1-ol, 2-methyl propane-2-ol
Arrange the following in the increasing order of their boiling point and give a reason for your ordering.
Propan-1-ol, propan-1, 2, 3-triol, propan-1, 3-diol, propan-2-ol
Can we use nucleophiles such as NH3, CH3O for the Nucleophilic substitution of alcohols?
Is it possible to oxidise t-butyl alcohol using acidified dichromate to form a carbonyl compound?
What happens when 1-phenyl ethanol is treated with acidified KMnO4.
Write the mechanism of acid catalysed dehydration of ethanol to give ethene.
How is phenol prepared from chloro benzene?
How is phenol prepared from isopropyl benzene?
Explain Kolbe’s reaction.
Write the chemical equation for Williamson's synthesis of 2-ethoxy-2-methyl pentane starting from ethanol and 2-methyl pentan-2-ol.
Write the structure of the aldehyde, carboxylic acid and ester that yield 4-methylpent-2-en-1-ol.
What is metamerism?
Give the structure and IUPAC name of metamers of 2-methoxy propane.
How is the conversion effected benzyl chloride to benzyl alcohol?
How is the conversion effected benzyl alcohol to benzoic acid?
Complete the following reaction.
\[\ce{CH3 - CH2 - OH ->[P Br3] A ->[aq. NaOH] B ->[Na] C}\]
Complete the following reaction.
\[\ce{C6H5 - OH ->[Zn dust] A ->[CH3Cl][Anhydrous AlCl3] B ->[acid KMnO4] C}\]
Complete the following reaction.
\[\ce{Anisole ->[{t}-butylchloride][AlCl3] A ->[Cl2/FeCl3] B ->[HBr] C}\]
Complete the following reaction.
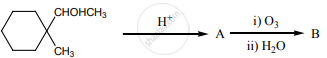
0.44 g of a monohydric alcohol, when added to methyl magnesium iodide in ether, liberates at STP 112 cm3 of methane with PCC the same alcohol form a carbonyl compound that answers the silver mirror test. Identify the compound.
Complete the following reaction.
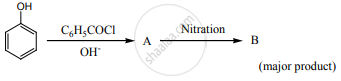
Complete the following reaction.
\[\ce{C6H5 - CH2CH(OH)CH(CH3)2 ->[conc. H2SO4]}\]
Phenol is distilled with Zn dust followed by Friedel-crafts alkylation with propyl chloride to give a compound B, B on oxidation gives (c) Identify A, B and C.
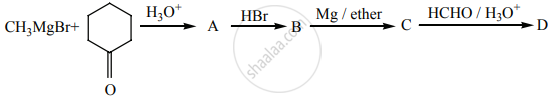
Identify A, B, C, D and write the complete equation.
What will be the product (X and A) for the following reaction?
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O^+] X ->[acid K2Cr2O7] A}\]
How will you convert acetylene into n-butyl alcohol?
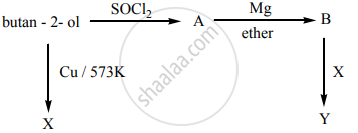
Predict the product A, B, X and Y in the following sequence of reaction.
3, 3-dimethyl butane-2-ol on treatment with conc. H2SO4 to give tetramethyl ethylene as a major product. Suggest a suitable mechanism.
Solutions for 11: Hydroxy Compounds and Ethers
![Samacheer Kalvi solutions for Chemistry - Volume 1 and 2 [English] Class 12 TN Board chapter 11 - Hydroxy Compounds and Ethers Samacheer Kalvi solutions for Chemistry - Volume 1 and 2 [English] Class 12 TN Board chapter 11 - Hydroxy Compounds and Ethers - Shaalaa.com](/images/chemistry-volume-1-and-2-english-class-12-tn-board_6:5f2b1b2038084cf381bfa42c826a928c.jpg)
Samacheer Kalvi solutions for Chemistry - Volume 1 and 2 [English] Class 12 TN Board chapter 11 - Hydroxy Compounds and Ethers
Shaalaa.com has the Tamil Nadu Board of Secondary Education Mathematics Chemistry - Volume 1 and 2 [English] Class 12 TN Board Tamil Nadu Board of Secondary Education solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. Samacheer Kalvi solutions for Mathematics Chemistry - Volume 1 and 2 [English] Class 12 TN Board Tamil Nadu Board of Secondary Education 11 (Hydroxy Compounds and Ethers) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. Samacheer Kalvi textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in Chemistry - Volume 1 and 2 [English] Class 12 TN Board chapter 11 Hydroxy Compounds and Ethers are Classification of Alcohols, The IUPAC System of Nomenclature, Reactions of Benzene Ring, Preparation of Ethers.
Using Samacheer Kalvi Chemistry - Volume 1 and 2 [English] Class 12 TN Board solutions Hydroxy Compounds and Ethers exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in Samacheer Kalvi Solutions are essential questions that can be asked in the final exam. Maximum Tamil Nadu Board of Secondary Education Chemistry - Volume 1 and 2 [English] Class 12 TN Board students prefer Samacheer Kalvi Textbook Solutions to score more in exams.
Get the free view of Chapter 11, Hydroxy Compounds and Ethers Chemistry - Volume 1 and 2 [English] Class 12 TN Board additional questions for Mathematics Chemistry - Volume 1 and 2 [English] Class 12 TN Board Tamil Nadu Board of Secondary Education, and you can use Shaalaa.com to keep it handy for your exam preparation.