Advertisements
Online Mock Tests
Chapters
2: p-Block Elements - I
3: p-Block Elements - II
4: Transition and Inner Transition Elements
5: Coordination Chemistry
6: Solid State
7: Chemical Kinetics
8: Ionic Equilibrium
9: Electro Chemistry
10: Surface Chemistry
11: Hydroxy Compounds and Ethers
12: Carbonyl Compounds and Carboxylic Acids
13: Organic Nitrogen Compounds
▶ 14: Biomolecules
15: Chemistry in Everyday Life
![Samacheer Kalvi solutions for Chemistry - Volume 1 and 2 [English] Class 12 TN Board chapter 14 - Biomolecules Samacheer Kalvi solutions for Chemistry - Volume 1 and 2 [English] Class 12 TN Board chapter 14 - Biomolecules - Shaalaa.com](/images/chemistry-volume-1-and-2-english-class-12-tn-board_6:5f2b1b2038084cf381bfa42c826a928c.jpg)
Advertisements
Solutions for Chapter 14: Biomolecules
Below listed, you can find solutions for Chapter 14 of Tamil Nadu Board of Secondary Education Samacheer Kalvi for Chemistry - Volume 1 and 2 [English] Class 12 TN Board.
Samacheer Kalvi solutions for Chemistry - Volume 1 and 2 [English] Class 12 TN Board 14 Biomolecules Evaluation [Pages 268 - 271]
Choose the correct answer:
Which one of the following rotates the plane polarized light towards left?
D(+) Glucose
L(+) Glucose
D(−) Fructose
D(+) Galactose
The correct corresponding order of names of four aldoses with configuration given below Respectively is:
L-Erythrose, L-Threose, L-Erythrose, D-Threose
D-Threose, D-Erythrose, L-Threose, L-Erythrose
L-Erythrose, L-Threose, D-Erythrose, D-Threose
D-Erythrose, D-Threose, L-Erythrose, L-Threose
Which one given below is a non-reducing sugar?
Glucose
Sucrose
maltose
Lactose
\[\ce{Glucose ->[(HCN)] Product ->[(hydrolysis)] Product ->[(HI + Heat)] A}\], the compound A is:
Heptanoic acid
2-Iodohexane
Heptane
Heptanol
Assertion: A solution of sucrose in water is dextrorotatory. But on hydrolysis in the presence of little hydrochloric acid, it becomes levorotatory.
Reason: Sucrose hydrolysis gives equal amounts of glucose and fructose. As a result of this change in sign of rotation is observed.
If both accretion and reason are true and reason is the correct explanation of assertion.
If both assertion and reason are true but reason is not the correct explanation of assertion.
If assertion is true but reason is false.
If both assertion and reason are false.
The central dogma of molecular genetics states that the genetic information flows from:
\[\ce{Amino acids -> Protein -> DNA}\]
\[\ce{DNA -> Carbohydrates -> Proteins}\]
\[\ce{DNA -> RNA -> Proteins}\]
\[\ce{DNA -> RNA -> Carbohydrates}\]
In a protein, various amino acids linked together by ____________.
Peptide bond
Dative bond
α-Glycosidic bond
β-Glycosidic bond
Among the following the achiral amino acid is:
2-ethylalanine
2-methylglycine
2-hydroxymethylserine
Tryptophan
The correct statement regarding RNA and DNA respectively is ____________.
the sugar component in RNA is an arabinose and the sugar component in DNA is ribose
the sugar component in RNA is 2’-deoxyribose and the sugar component in DNA is arabinose
the sugar component in RNA is an arabinose and the sugar component in DNA is 2’-deoxyribose
the sugar component in RNA is ribose and the sugar component in DNA is 2’-deoxyribose
In aqueous solution of amino acids mostly exists in, ____________.
NH2–CH(R)–COOH
NH2–CH(R)–COO–
H3N+–CH(R)–COOH
H3N+–CH(R)–COO–
Which one of the following is not produced by body?
DNA
Enzymes
Hormones
Vitamins
The number of sp2 and sp3 hybridised carbon in fructose are respectively ____________.
1 and 4
4 and 2
5 and 1
1 and 5
Vitamin B2 is also known as ____________.
Riboflavin
Thiamine
Nicotinamide
Pyridoxine
The pyrimidine bases present in DNA are ____________.
Cytosine and Adenine
Cytosine and Guanine
Cytosine and Thiamine
Cytosine and Uracil
Among the following L-serine is:
The secondary structure of a protein refers to ____________.
fixed configuration of the polypeptide backbone
hydrophobic interaction
sequence of α-amino acids
α-helical backbone
Which of the following vitamins is water soluble?
Vitamin E
Vitamin K
Vitamin A
Vitamin B
Complete hydrolysis of cellulose gives ____________.
L-Glucose
D-Fructose
D-Ribose
D-Glucose
Which of the following statement is not correct?
Ovalbumin is a simple food reserve in egg-white.
Blood proteins thrombin and fibrinogen are involved in blood clotting.
Denaturation makes protein more active.
Insulin maintains the sugar level in the human body.
Glucose is an aldose. Which one of the following reactions is not expected with glucose?
It does not form oxime.
It does not react with Grignard reagent.
It does not form osazones.
It does not reduce tollens reagent.
If one strand of the DNA has the sequence ‘ATGCTTGA’, then the sequence of complementary strand would be ____________.
TACGAACT
TCCGAACT
TACGTACT
TACGRAGT
Insulin, a hormone chemically is ___________.
Fat
Steroid
Protein
Carbohydrates
α-D (+) Glucose and β-D (+) glucose are ____________.
Epimers
Anomers
Enantiomers
Conformational isomers
Which of the following are epimers?
D(+)-Glucose and D(+)-Galactose
D(+)-Glucose and D(+)-Mannose
Neither D(+)-Glucose and D(+)-Galactose nor D(+)-Glucose and D(+)-Mannose
Both D(+)-Glucose and D(+)-Galactose and D(+)-Glucose and D(+)-Mannose
Which of the following amino acids is achiral?
Alanine
Leucine
Proline
Glycine
Short Answer Questions:
What type of linkages holds together monomers of DNA?
Give the differences between the primary and secondary structure of proteins.
Name the vitamin whose deficiency causes rickets.
Name the vitamin whose deficiency causes scurvy.
Write the Zwitter ion structure of alanine.
Give any three differences between DNA and RNA.
Write a short note on peptide bond.
Give two differences between Hormones and vitamins.
Write a note on denaturation of proteins.
What are reducing and non-reducing sugars?
Why carbohydrates are generally optically active?
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Starch
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Fructose
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Sucrose
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Lactose
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Maltose
How are vitamins classified?
What are hormones? Give examples.
Write the structure of all possible dipeptides which can be obtained from glycine and alanine.
Define enzymes
Write the structure of α-D (+) glucopyranose.
What are different types of RNA which are found in cells?
Write a note on the formation of α-helix.
What are the functions of lipids in living organisms?
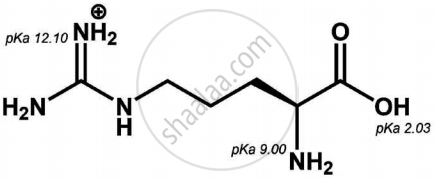
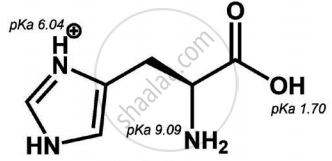
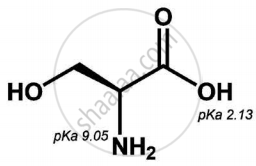
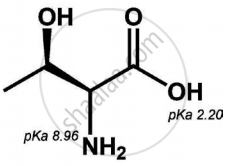
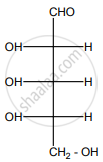
Is the following sugar, D-sugar or L-sugar?
Solutions for 14: Biomolecules
![Samacheer Kalvi solutions for Chemistry - Volume 1 and 2 [English] Class 12 TN Board chapter 14 - Biomolecules Samacheer Kalvi solutions for Chemistry - Volume 1 and 2 [English] Class 12 TN Board chapter 14 - Biomolecules - Shaalaa.com](/images/chemistry-volume-1-and-2-english-class-12-tn-board_6:5f2b1b2038084cf381bfa42c826a928c.jpg)
Samacheer Kalvi solutions for Chemistry - Volume 1 and 2 [English] Class 12 TN Board chapter 14 - Biomolecules
Shaalaa.com has the Tamil Nadu Board of Secondary Education Mathematics Chemistry - Volume 1 and 2 [English] Class 12 TN Board Tamil Nadu Board of Secondary Education solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. Samacheer Kalvi solutions for Mathematics Chemistry - Volume 1 and 2 [English] Class 12 TN Board Tamil Nadu Board of Secondary Education 14 (Biomolecules) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. Samacheer Kalvi textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in Chemistry - Volume 1 and 2 [English] Class 12 TN Board chapter 14 Biomolecules are Carbohydrates, Proteins, Lipids, Introduction of Vitamins, Nucleic Acids, Chemical Coordination.
Using Samacheer Kalvi Chemistry - Volume 1 and 2 [English] Class 12 TN Board solutions Biomolecules exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in Samacheer Kalvi Solutions are essential questions that can be asked in the final exam. Maximum Tamil Nadu Board of Secondary Education Chemistry - Volume 1 and 2 [English] Class 12 TN Board students prefer Samacheer Kalvi Textbook Solutions to score more in exams.
Get the free view of Chapter 14, Biomolecules Chemistry - Volume 1 and 2 [English] Class 12 TN Board additional questions for Mathematics Chemistry - Volume 1 and 2 [English] Class 12 TN Board Tamil Nadu Board of Secondary Education, and you can use Shaalaa.com to keep it handy for your exam preparation.