Advertisements
Advertisements
प्रश्न
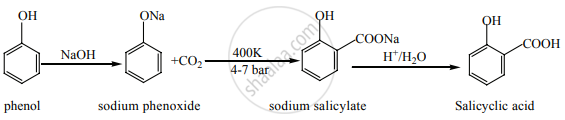
Explain Kolbe’s reaction.
उत्तर
Kolbe’s (or) Kolbe’s Schmit reaction:
In this reaction, phenol is first converted into sodium phenoxide which is more reactive than phenol towards electrophilic substitution reaction with CO2. Treatment of sodium phenoxide with CO2 at 400 K, 4-7 bar pressure followed by acid hydrolysis gives salicylic acid.
APPEARS IN
संबंधित प्रश्न
Assertion: Phenol is more acidic than ethanol.
Reason: Phenoxide ion is resonance stabilized.
Which of the following compound can be used as antifreeze in automobile radiators?
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroboration
Arrange the following in the increasing order of their boiling point and give a reason for your ordering.
Propan-1-ol, propan-1, 2, 3-triol, propan-1, 3-diol, propan-2-ol
Is it possible to oxidise t-butyl alcohol using acidified dichromate to form a carbonyl compound?
Write the structure of the aldehyde, carboxylic acid and ester that yield 4-methylpent-2-en-1-ol.
What will be the product (X and A) for the following reaction:
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O+] X ->[acid K2Cr2O7]A}\]
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in the following method.
Acid catalysed hydration
Predict the major product, when 2-methyl but – 2 – ene is converted into an alcohol in each of the following method.
Acid catalysed hydration.
