Advertisements
Advertisements
प्रश्न
Identify the product(s) is/are formed when 1-methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
उत्तर
\[\ce{\underset{1-methoxy propane}{CH3 - O - CH2 - CH2 - CH3} + HI -> \underset{\underset{(Iodomethane)}{Methyl Iodide}}{CH3I} + \underset{1-propane}{CH3 - CH2 - CH2 - OH} ->[HI] \underset{1-Iodopropane}{CH3 - CH2 - CH2 - I} + H2O}\]
Ethers having the primary alkyl group undergo an SN2 reaction.
APPEARS IN
संबंधित प्रश्न
Assertion: Phenol is more acidic than ethanol.
Reason: Phenoxide ion is resonance stabilized.
Assertion: Phenol is more reactive than benzene towards electrophilic substitution reaction
Reason: In the case of phenol, the intermediate arenium ion is more stabilized by resonance.
HO CH2 CH2 – OH on heating with periodic acid gives ____________.
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroboration
Arrange the following in the increasing order of their boiling point and give a reason for your ordering.
Butan-2-ol, Butan-1-ol, 2-methyl propane-2-ol
How is the conversion effected benzyl alcohol to benzoic acid?
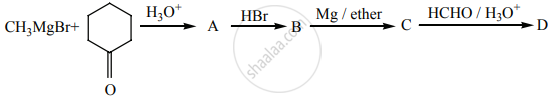
Identify A, B, C, D and write the complete equation.
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI
Identify the product(s) is/are formed when 1-methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in each of the following methods.
Acid catalysed hydration
