Advertisements
Advertisements
Question
Identify the product(s) is/are formed when 1-methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Solution
\[\ce{\underset{1-methoxy propane}{CH3 - O - CH2 - CH2 - CH3} + HI -> \underset{\underset{(Iodomethane)}{Methyl Iodide}}{CH3I} + \underset{1-propane}{CH3 - CH2 - CH2 - OH} ->[HI] \underset{1-Iodopropane}{CH3 - CH2 - CH2 - I} + H2O}\]
Ethers having the primary alkyl group undergo an SN2 reaction.
APPEARS IN
RELATED QUESTIONS
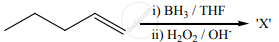
The X is
Which one of the following will react with phenol to give salicylaldehyde after hydrolysis.
Among the following ethers which one will produce methyl alcohol on treatment with hot HI?
Explain Kolbe’s reaction.
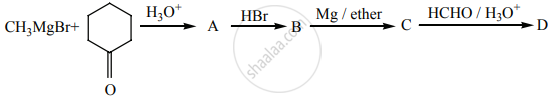
Identify A, B, C, D and write the complete equation.
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in the following method.
Acid catalysed hydration
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in each of the following methods.
Acid catalysed hydration
Predict the major product, when 2-methyl but – 2 – ene is converted into an alcohol in each of the following method.
Acid catalysed hydration.
