Advertisements
Advertisements
Question
Among the following ethers which one will produce methyl alcohol on treatment with hot HI?
Options
\[\ce{(H3C)3 C - O - CH3}\]
\[\ce{(CH3)2 - CH - CH2 - O - CH3}\]
\[\ce{CH3 (CH2)3 - O - CH3}\]
\[\begin{array}{cc}
\ce{CH3 - CH2 - CH - O - CH3}\\
|\\
\phantom{...}\ce{CH3}
\end{array}\]
Solution
\[\ce{(H3C)3 C - O - CH3}\]
APPEARS IN
RELATED QUESTIONS
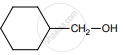 on treatment with Con. H2SO4 predominately gives
on treatment with Con. H2SO4 predominately gives
The reaction can be classified as
How is phenol prepared from isopropyl benzene?
3, 3-dimethyl butane-2-ol on treatment with conc. H2SO4 to give tetramethyl ethylene as a major product. Suggest a suitable mechanism.
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in each of the following method.
Acid catalysed hydration
Identify the product (s) is / are formed when 1 – methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in the following method.
Acid catalysed hydration
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
What will be the product (X and A)for the following reaction
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O] X ->[acid K2Cr2O7] A}\]
What will be the product (X and A)for the following reaction
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O^+]X ->[acid K2Cr2O7]A}\]
