Advertisements
Advertisements
Question
The reaction can be classified as
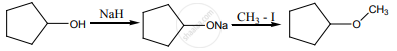
Options
dehydration
Williamson alcohol synthesis
Williamson ether synthesis
dehydrogenation of alcohol
Solution
Williamson ether synthesis
APPEARS IN
RELATED QUESTIONS
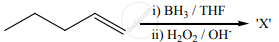
The X is
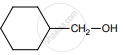 on treatment with Con. H2SO4 predominately gives
on treatment with Con. H2SO4 predominately gives
Assertion: Phenol is more reactive than benzene towards electrophilic substitution reaction
Reason: In the case of phenol, the intermediate arenium ion is more stabilized by resonance.
HO CH2 CH2 – OH on heating with periodic acid gives ____________.
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroboration
How is phenol prepared from chloro benzene?
0.44 g of a monohydric alcohol, when added to methyl magnesium iodide in ether, liberates at STP 112 cm3 of methane with PCC the same alcohol form a carbonyl compound that answers the silver mirror test. Identify the compound.
What will be the product (X and A)for the following reaction
acetylchloride \[\ce{{acetylchloride}->[i)CH3MgBr][ii)H3O+]X ->[acidK2Cr2O7] A}\]
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI
Identify the product (s) is / are formed when 1 – methoxy propane is heated with excess HI. Name the mechanism involved in the reaction
