Advertisements
Advertisements
Question
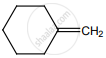
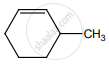
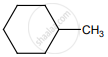
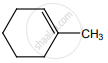
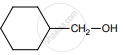 on treatment with Con. H2SO4 predominately gives
on treatment with Con. H2SO4 predominately gives
Options
Solution
APPEARS IN
RELATED QUESTIONS
Which of the following compounds on reaction with methyl magnesium bromide will give tertiary alcohol.
One mole of an organic compound (A) with the formula C3H8O reacts completely with two moles of HI to form X and Y. When Y is boiled with aqueous alkali it forms Z. Z answers the iodoform test. The compound (A) is ___________.
Complete the following reaction.
\[\ce{C6H5 - OH ->[Zn dust] A ->[CH3Cl][Anhydrous AlCl3] B ->[acid KMnO4] C}\]
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI
Identify the product (s) is / are formed when 1 – methoxy propane is heated with excess HI. Name the mechanism involved in the reaction
What will be the product (X and A) for the following reaction?
acetylchloride\[\ce{->[i) CH3MgBr][ii) H3O^+]X->[acid K2Cr2O7]A}\]
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
Identify the product(s) is/are formed when 1-methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
The correct IUPAC name of the compound,
\[\begin{array}{cc}
\ce{CH3}\phantom{......}\\
|\phantom{........}\\
\ce{H3C - CH - CH - CH - CH2 - OH}\\
|\phantom{............}|\phantom{........}\\
\ce{Cl}\phantom{...........}\ce{CH3}\phantom{......}
\end{array}\]