Advertisements
Advertisements
Question
One mole of an organic compound (A) with the formula C3H8O reacts completely with two moles of HI to form X and Y. When Y is boiled with aqueous alkali it forms Z. Z answers the iodoform test. The compound (A) is ___________.
Options
propan-2-ol
propan-1-ol
ethoxy ethane
methoxy ethane
Solution
One mole of an organic compound (A) with the formula C3H8O reacts completely with two moles of HI to form X and Y. When Y is boiled with aqueous alkali it forms Z. Z answers the iodoform test. The compound (A) is methoxy ethane.
APPEARS IN
RELATED QUESTIONS
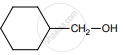 on treatment with Con. H2SO4 predominately gives
on treatment with Con. H2SO4 predominately gives
What is the major product obtained when two moles of ethyl magnesium bromide is treated with methyl benzoate followed by acid hydrolysis.
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroboration
Is it possible to oxidise t-butyl alcohol using acidified dichromate to form a carbonyl compound?
How is the conversion effected benzyl chloride to benzyl alcohol?
What will be the product (X and A) for the following reaction?
acetylchloride \[\ce{->[i) CH3MgBr][ii) H3O+] X ->[acid K2Cr2O7] A}\]
Identify the product (s) is / are formed when 1 – methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in the following method.
Acid catalysed hydration
What will be the product (X and A)for the following reaction
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O] X ->[acid K2Cr2O7] A}\]
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in each of the following methods.
Acid catalysed hydration
