Advertisements
Advertisements
Question
What is the major product obtained when two moles of ethyl magnesium bromide is treated with methyl benzoate followed by acid hydrolysis.
Solution

APPEARS IN
RELATED QUESTIONS
Which of the following compounds on reaction with methyl magnesium bromide will give tertiary alcohol.
\[\ce{(CH3)3 - C - CH(OH) CH3 ->[con H2SO4] X (major product)}\]
Assertion: Phenol is more acidic than ethanol.
Reason: Phenoxide ion is resonance stabilized.
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroxylation using bayers reagent
Explain Kolbe’s reaction.
0.44 g of a monohydric alcohol, when added to methyl magnesium iodide in ether, liberates at STP 112 cm3 of methane with PCC the same alcohol form a carbonyl compound that answers the silver mirror test. Identify the compound.
Complete the following reaction.
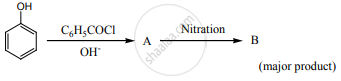
Complete the following reaction.
\[\ce{C6H5 - CH2CH(OH)CH(CH3)2 ->[conc. H2SO4]}\]
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI
What will be the product (X and A) for the following reaction?
acetylchloride \[\ce{->[i) CH3MgBr][ii) H3O+] X ->[acid K2Cr2O7] A}\]
