Advertisements
Advertisements
प्रश्न
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroboration
उत्तर
\[\begin{array}{cc}
\phantom{...............................................}\ce{CH3}\\
\phantom{............................................}|\\
\ce{6CH3 - C = CH - CH3 + B2H6 -> 2[(CH3)2 CH - CH -]_3 B}\\
|\phantom{..........................................}\\
\ce{\underset{(2-methyl-but-2-ene)}{CH3}}\phantom{.......................................}
\end{array}\]
\[\begin{array}{cc}
\phantom{..}\ce{CH3}\phantom{............................}\ce{CH3}\phantom{...}\ce{CH3}\phantom{...}\\
\phantom{.}|\phantom{................................}|\phantom{......}|\phantom{....}\\
\ce{[(CH3)2 CH - CH -]_3 B + 3H2O2 ->[OH^Θ] \underset{(3-methyl-butan-2-ol)}{3CH3 - CH - CH - OH} + H3BO3}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
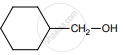 on treatment with Con. H2SO4 predominately gives
on treatment with Con. H2SO4 predominately gives
Carbolic acid is ____________.
Assertion: Phenol is more reactive than benzene towards electrophilic substitution reaction
Reason: In the case of phenol, the intermediate arenium ion is more stabilized by resonance.
Among the following ethers which one will produce methyl alcohol on treatment with hot HI?
Explain Kolbe’s reaction.
What will be the product (X and A) for the following reaction:
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O+]X->[acid K2Cr2O7]A}\]
Identify the product (s) is / are formed when 1 – methoxy propane is heated with excess HI. Name the mechanism involved in the reaction
What will be the product (X and A)for the following reaction
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O] X ->[acid K2Cr2O7] A}\]
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
What will be the product (X and A)for the following reaction
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O^+]X ->[acid K2Cr2O7]A}\]
