Advertisements
Advertisements
प्रश्न
Select the correct answer
Which of the following electronic structure is of a metal?
पर्याय
2, 8, 8
2, 7
2, 8, 2
2, 8, 4
उत्तर
2, 8, 2 because it has only 2 electrons in its valence shell which can be lost to form a dipositive cation.
APPEARS IN
संबंधित प्रश्न
The electronic configuration of an element T is 2, 8, 8, 1.
Is it a metal or a non-metal?
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 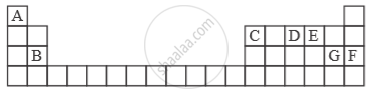
Which non-metallic element has the valency of 2?
Among the elements of the second period, Li to Ne, pick out the element that is the most reactive metal
In the third period, which is the most metallic and most non-metallic element?
Explain
Larger the atomic size, more metallic is the element.
Match the atomic number with the following:
A metal of valency one.
The tendency of an element to form cation is the _______ character of that element.
The lightest liquid metal is ______
Three elements B, Si and Ge are
Identify the elements with the following property and arrange them in increasing order of their reactivity
- An element which is a soft and reactive metal
- The metal which is an important constituent of limestone
- The metal which exists in liquid state at room temperature
