Advertisements
Advertisements
प्रश्न
Show the heterolysis of a covalent bond by using curved arrow notation and complete the following equation. Identify the nucelophile.
CH3 – Br + KOH →
उत्तर
\[\ce{CH3 – Br + KOH →}\]
\[\ce{CH3 – Br + KQH → CH3OH + KBr}\]
Nucleophile is: OH–
APPEARS IN
संबंधित प्रश्न
For the following reactions
- \[\ce{CH3CH2CH2Br + KOH → CH3 – CH = CH2 + KBr + H2O}\]
- \[\ce{(CH3)3CBr + KOH → (CH3)3COH + KBr}\]
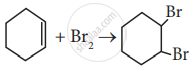
Which of the following statement is correct?
What is the hybridisation state of benzyl carbonium ion?
Which of the following species is not electrophilic in nature?
Hyper Conjugation is also known as
Heterolytic fission of C–C bond results in the formation of ______.
Which of the following represent a set of nucleophiles?
The geometrical shape of carbocation is ______.
Write a short note on hyperconjucation.
Show the heterolysis of a covalent bond by using curved arrow notation and complete the following equation. Identify the nucelophile.
\[\ce{CH3 – O – CH3 + HI →}\]
Explain the electromeric effect.
