Advertisements
Advertisements
प्रश्न
Some of the properties of water are described below. Which of them is/are not correct?
(i) Water is known to be a universal solvent.
(ii) Hydrogen bonding is present to a large extent in liquid water.
(iii) There is no hydrogen bonding in the frozen state of water.
(iv) Frozen water is heavier than liquid water.
उत्तर
(iii) There is no hydrogen bonding in the frozen state of water.
(iv) Frozen water is heavier than liquid water.
Explanation:
The presence of extensive hydrogen bonding between water molecules gives it remarkable features in the condensed phase (liquid and solid phases). In comparison to \[\ce{H2S}\] and \[\ce{H2Se}\], this results in a high freezing point, high boiling point, high heat of vaporisation, and high heat of fusion.
APPEARS IN
संबंधित प्रश्न
Sea water is a bad conductor of electricity.
Explain the picture in your own words.
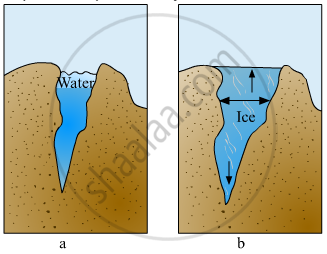
Explain the various properties of water.
Which of the following equation depicts reducing nature of \[\ce{H2O2}\]?
If same mass of liquid water and a piece of ice is taken, then why is the density of ice less than that of liquid water?
Melting point, enthalpy of vapourisation and viscosity data of \[\ce{H2O}\] and \[\ce{D2O}\] is given below :
| \[\ce{H, O}\] | \[\ce{D2O}\] | |
| Melting point / K | 373.0 | 374.4 |
| Enthalpy of vapourisation at (373 K)/kJ mol–1 | 40.66 | 41.61 |
| Viscosity/centipoise | 0.8903 | 1.107 |
On the basis of this data explain in which of these liquids intermolecular forces are stronger?
Explain why \[\ce{HCl}\] is a gas and \[\ce{HF}\] is a liquid.
Freezing of water will cause an ______ is the volume.
Water is circulated around the car engine using the ______ pump and the heat is absorbed.
The water having more dissolved O2 is ______.
