Advertisements
Advertisements
प्रश्न
State whether the following statement is true or false:
A chemical equation can be balanced easily by altering the formula of a reactant or product.
उत्तर
False
Formula of a reactant or product cannot be altered, but chemical equation is balanced by multiplying the reactant or product with numbers like 2,3,4,5 and so on to balance the number of atoms in the chemical equation.
APPEARS IN
संबंधित प्रश्न
Translate the following statement into chemical equation and then balance it.
Hydrogen gas combines with nitrogen to form ammonia
Give one example of a chemical reaction.
Correct and balance the following equation:
Ca + H2O  CaOH + H
CaOH + H
Fill in the following blank with suitable word:
A solution made in water is known as an ........... solution and indicated by the symbol ...........
You are given the solution of lead nitrate. In order to obtain a yellow precipitate you should mix with it a solution of:
(a) potassium chloride
(b) potassium nitride
(c) potassium sulphide
(d) potassium iodide
Balance the following chemical equation:
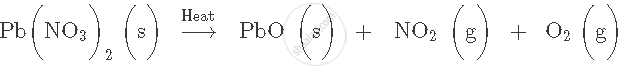
Write word equation for the following molecular equation:
\[\ce{NH3 [g] + HCl [g] ⇌ NH4Cl [s]}\]
Word equation:
State which characteristic is observed in the above reaction.
Give an-example of a similar above characteristic, seen between two other gaseous reactants.
In certain reaction an insoluble solid called precipitate is formed. State the colour and name of the precipitate formed in the following reaction involving addition of:
Iron [III] chloride to ammonium hydroxide.
Which of the following is not a physical change?
Complete the following blank in the equation as indicated.
\[\ce{CaH2_{(s)} + 2H2O_{( aq)}-> Ca(OH)2_{(s)} + 2H2_{(g)}}\]
Molecules: 6.02 × 1023 + ______ `→` ______ + ______
