Advertisements
Advertisements
प्रश्न
Translate the following statement into chemical equation and then balance it.
Hydrogen gas combines with nitrogen to form ammonia
उत्तर
\[\ce{H2_{(g)} + N2_{(g)} -> NH3_{(g)}}\]
Balanced equation:
\[\ce{3H2_{(g)} + N2_{(g)} -> 2NH3_{(g)}}\]
APPEARS IN
संबंधित प्रश्न
Write the balanced chemical equation for the following reaction.
\[\ce{Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water}\]
How will you indicate Endothermic reaction in a chemical equation?
Give one example of a chemical reaction.
Balance the given equation:
AI(OH)3  AI2O3 +H2O
AI2O3 +H2O
Balance the following chemical equation:

Express the following facts in the form of a balanced chemical equation:
"When a strip of copper metal is placed in a solution of silver nitrate, metallic silver is precipitated and a solution containing copper nitrate is formed".
A colourless lead salt, when heated, produces a yellow residue and brown fumes.
(a) Name the lead salt.
(b) Name the brown fumes.
(c) Write a chemical equation of the reaction involved.
Balance the following equation Also name the product formed.
`"K" + "CI"_2 → "KCI"`
While performing the experiment with raisins to determine the percentage of water absorbed by them, a student made following measurements:
Mass of water in the beaker = 40 g
Mass of raisins before soaking = 5 g
Mass of raisins after soaking for 2 hours = 8 g
Mass of water left in the beaker after the experiment = 35 g
The percentage of water absorbed by raisins is:
What information do the following chemical equations convey? Mg + 2HCl → MgCl2+ H2
Match column A with column B.
| Column A | Column B |
| (a) Blue salt changes to white and then black | (i) Ammonium dichromate |
| (b) Orange coloured compound changes to green. | (ii) Iodine |
| (c) Red compound changes to brown and then yellow. | (iii) Zinc Nitrate |
| (d) White to yellow when hot and white when cold. | (iv) Copper sulphate |
| (e) Violet solid changes to violet vapours. | (v) Red Lead |
A student mixes sodium sulphate powder in barium chloride powder. What change would the student observe on mixing the two powders? Justify your answer and explain how he can obtain the desired change.
In certain reaction a change of state is observed i.e. solid to liquid, liquid to gas etc. – State the change of state of the products – to give the respective reactant.
\[\ce{2H2O ->[electric][current] 2H2 + O2}\]
Name the following:
The gas evolved when a dilute acid is added to chalk [limestone].
Balance the following simple equation:
\[\ce{Ca(OH)2 + HNO3 -> Ca(NO3)2 + H2O}\]
Write a balanced equation for the following word equation:
Potassium bicarbonate → Potassium carbonate + Water + Carbon dioxide
Balance the given chemical reaction as per the instructions below.
\[\ce{NaOH + H2SO4 -> Na2SO4 + H2O}\]
Write names of reactants and products of chemical reaction.
The chemical equation Na2SO4(aq) + BaCl2(aq) → BaSO4(s)↓ + 2NaCl(aq) represents which of the following types of reaction?
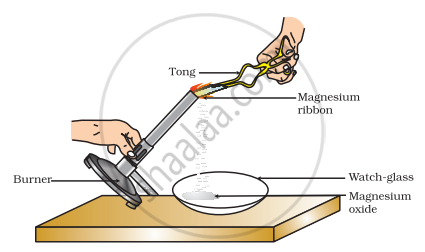
Which of the following is the correct observation of the reaction shown in the above set up?
A student took a small amount of copper oxide in a conical flask and added dilute hydrochloric acid to it with constant stirring. He observed a change in colour of the solution.
- Write the name of the compound formed and its colour.
- Write a balanced chemical equation for the reaction involved.
