Advertisements
Advertisements
प्रश्न
Sulphuric acid reacts with sodium hydroxide as follows:
\[\ce{H2SO4 + 2NaOH -> Na2SO4 + 2H2O}\]
When 1 L of 0.1 M sulphuric acid solution is allowed to react with 1 L of 0.1 M sodium hydroxide solution, the amount of sodium sulphate formed and its molarity in the solution obtained is:
(i) 0.1 mol L–1
(ii) 7.10 g
(iii) 0.025 mol L–1
(iv) 3.55 g
उत्तर
(ii) 7.10 g
(iii) 0.025 mol L–1
Explanation:
0.1 mole \[\ce{H2SO4}\] reacts with 1 mole of \[\ce{NaOH}\].
0.1 mole of \[\ce{NaOH}\] will react with `0.1/2` mole of \[\ce{H2SO4}\].
Here, \[\ce{NaOH}\] is the limiting reagent.
2 mole of \[\ce{NaOH}\] produces 1 mole of \[\ce{Na2SO4}\].
0.1 mole of \[\ce{NaOH}\] will give `0.1/2` mole of \[\ce{Na2SO4}\].
No. of mole = `"Given mass"/("Molar mass" (Na_2SO_4))`
On substituting the value in tha above equation, the mass can be calculated as 0.05 mol = `"Given mass"/(142 g mol^-1)`
Given mass = 7.10 g
Volume of solution after mixing is 2 L.
So, the molarity of \[\ce{Na2SO4}\] is,
Molarity = `0.05/2` = 0.025 mol L–1
APPEARS IN
संबंधित प्रश्न
Calculate the amount of carbon dioxide that could be produced when 1 mole of carbon is burnt in air.
Explain the term mole fraction
Solve the following problem:
Find out the molar mass of the following compounds:
Copper sulphate crystal (CuSO4.5H2O)
(At. mass: Cu = 63.5; S = 32; O = 16; H = 1; Na = 23; C = 12; Fe = 56; N = 14)
Solve the following problem:
Perform the following calculation. Round off your answer to two digits.
`33/(9.00xx10^-4)`
Solve the following problem:
A 1.000 mL sample of acetone, a common solvent used as a paint remover, was placed in a small bottle whose mass was known to be 38.0015 g.
The following values were obtained when the acetone - filled bottle was weighed: 38.7798 g, 38.7795 g and 38.7801 g. How would you characterise the precision and accuracy of these measurements if the actual mass of the acetone was 0.7791 g?
Solve the following problem:
The hourly energy requirements of an astronaut can be satisfied by the energy released when 34 grams of sucrose are “burnt” in his body. How many grams of oxygen would be needed to be carried in space capsule to meet his requirement for one day?
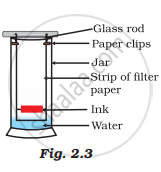
A child wanted to separate the mixture of dyes constituting a sample of ink. He marked a line by the ink on the filter paper and placed the filter paper in a glass containing water as shown in Fig.2.3. The filter paper was removed when the water moved near the top of the filter paper.
(i) What would you expect to see, if the ink contains three different coloured components?
(ii) Name the technique used by the child.
(iii) Suggest one more application of this technique.
Calculate the mass of sodium sulphate required to prepare its 20% (mass percent) solution in 100g of water?
If the concentration of glucose \[\ce{(C6H12O6)}\] in blood is 0.9 g L–1, what will be the molarity of glucose in blood?
What will be the molality of the solution containing 18.25 g of \[\ce{HCl}\] gas in 500 g of water?
