Advertisements
Advertisements
प्रश्न
Taking into consideration the relationship in the first pair, complete the second pair
2H2 + O2 → 2H2O :Combination Reaction :: 2HgO → 2Hg + O2:_________
उत्तर
Decomposition reaction
APPEARS IN
संबंधित प्रश्न
Write one equation for decomposition reactions where energy is supplied in the form of heat.
The white solid compound A decomposes quite rapidly on heating in the presence of a black substance X to form a solid compound B and a gas C. When an aqueous solution of compound B is reacted with silver nitrate solution, then a white precipitate of silver chloride is obtained along with potassium nitrate solution. Gas C does not burn itself but helps burn other things.
(a) What is compound A?
(b) What is compound B?
(c) What is gas C?
(d) What do you think is the black substance X? What is its function?
(e) What is the general name of substances like X?
Give one example of a decomposition reaction which is carried out by applying heat.
Give one example of a decomposition reaction which is carried out with electricity.
What type of chemical reaction is used to extract metals from their naturally occurring compounds like oxides or chlorides?
What type of reaction is represented by the following equation?
2 FeSO4 → Fe2 O3 + SO2 + SO3
What type of reaction is represented by the following equation?
NH4 CI → NH3 + HCI
What type of chemical reaction take place when electricity is passed through water?
What is the colour of ferrous sulphate crystals? How does this colour change after heating?
What are thermal decomposition reactions ? Explain with an example.
What do you mean by redox reaction ? Explain with the help of an example.
What does one mean by endothermic reaction? Give example.
Identify the type of following reaction :
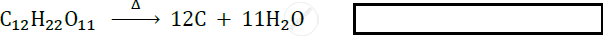
(a) Design an activity to demonstrate the decomposition reaction of lead nitrate.
(b) Draw labelled diagram of the experimental set-up. List two main observations.
(c) Write balanced chemical equation for the reaction stating the physical state of the reactant and the products.
A student wants to study a decomposition reaction by taking ferrous sulphate crystals. Write two precautions he must observe while performing the experiment.
Classify the following reaction into different type:
\[\ce{2KClO3(s)->[\Delta] 2KCl(aq) + 3O2(g)}\]
Differentiate between direct combination reaction and a decomposition reaction.
Classify the following reaction into –
- Direct combination
- Decomposition
- Displacement
- Double decomposition
The reaction is – Zinc hydroxide on heating gives zinc oxide and water.
Give a balanced equation for –
An electrolytic decomposition reaction involving a neutral liquid
Give a balanced equation for –
A double decomposition neutralization reaction involving an acid and a base
Give a balanced equation for the following type of reaction:
A thermal decomposition reaction in which a metallic nitrate decomposes to give – a basic oxide.
Give a balanced equation for the following type of reaction:
A thermal decomposition reaction in which a compound decomposes to give two new compounds.
Differentiate between the following:
Electrolytic decomposition and photochemical decomposition.
Electrolysis of water is a decomposition reaction. The mole ratio of hydrogen and oxygen gases liberated during electrolysis of water is:
Which one of the following processes involves chemical reactions?
On heating blue coloured powder of copper (II) nitrate, in a boiling tube, copper oxide (black), oxygen gas and a brown gas X is formed
- Write a balanced chemical equation of the reaction.
- Identify the brown gas X evolved.
- Identify the type of reaction.
- What could be the pH range of aqueous solution of the gas X?
Complete the following reaction:
\[\ce{C_12H_22O11->[Heat]}\] ______ + ______.
A metal nitrate 'A' on heating gives a metal oxide along with evolution of a brown coloured gas 'B' and a colourless gas, which helps in burning. Aqueous solution of 'A' when reacted with potassium iodide forms a yellow precipitate.
- Identify 'A' and 'B'
- Name the types of the reactions involved in the above statement.
What is observed when silver chloride is exposed to sunlight? Give the type of reaction involved.
When lead nitrate is heated strongly in a boiling tube, two gases are liberated and a solid residue is left behind in the test tube.
- Name the type of chemical reaction and define it.
- Write the name and formula of the coloured gas liberated.
- Write the balanced chemical equation for the reaction.
- Name the residue left in the test tube and state the method of testing its nature (acidic/basic).
Write the molecular formula of calcium carbonate.
