Advertisements
Advertisements
प्रश्न
Taking the Bohr radius as a0 = 53 pm, the radius of Li++ ion in its ground state, on the basis of Bohr’s model, will be about ______.
पर्याय
53 pm
27 pm
18 pm
13 pm
उत्तर
Taking the Bohr radius as a0 = 53 pm, the radius of Li++ ion in its ground state, on the basis of Bohr’s model, will be about 18 pm.
Explanation:
Bohr’s radius of orbit (for Hydrogen and H,-like atoms): For an electron around a stationary nucleus, the electrostatic force of attraction provides the necessary centripetal force.
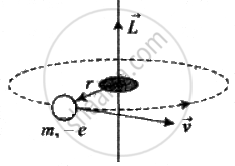
i.e., `1/(4πε_0) ((Ze)e)/r^2 = (mv^2)/r` ......(i)
Also `mvr = (nh)/(2pi)` ......(ii)
From equations (i) and (ii), the radius of nth orbit
`r_n = (n^2h^2)/(4pi^2 kZme^2) = (n^2h^2ε_0)/(pimZe^2) = 0.53 n^2/Z Å` ......`(k = 1/(4πε_0))`
⇒ `r_n ∝ n^2/Z` or `r_n ∝ 1/Z`
`r_n = a_0 n^2/Z`, where a0 = the Bhor radius = 53 pm
‘The atomic number (Z) of lithium is 3.
As `r_n = a_0 n^2/Z`,
Therefore, the radius of Li++ ion in its ground state, on the basis of Bohr’s model. will be about `1/3` times to that of Bohr's radius.
Therefore, the radius of lithium ion is near r = `53/3` = 18 pm.
APPEARS IN
संबंधित प्रश्न
(i) State Bohr's quantization condition for defining stationary orbits. How does the de Broglie hypothesis explain the stationary orbits?
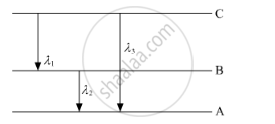
(ii) Find the relation between three wavelengths λ1, λ2 and λ3 from the energy-level diagram shown below.
The numerical value of ionization energy in eV equals the ionization potential in volts. Does the equality hold if these quantities are measured in some other units?
In a laser tube, all the photons
A beam of light having wavelengths distributed uniformly between 450 nm to 550 nm passes through a sample of hydrogen gas. Which wavelength will have the least intensity in the transmitted beam?
A beam of monochromatic light of wavelength λ ejects photoelectrons from a cesium surface (Φ = 1.9 eV). These photoelectrons are made to collide with hydrogen atoms in ground state. Find the maximum value of λ for which (a) hydrogen atoms may be ionized, (b) hydrogen atoms may get excited from the ground state to the first excited state and (c) the excited hydrogen atoms may emit visible light.
A filter transmits only the radiation of wavelength greater than 440 nm. Radiation from a hydrogen-discharge tube goes through such a filter and is incident on a metal of work function 2.0 eV. Find the stopping potential which can stop the photoelectrons.
According to Bohr's model of hydrogen atom, an electron can revolve round a proton indefinitely, if its path is ______.
What is the energy associated with first orbit of Li2+ (RH = 2.18 × 10-18)?
The wavelength of the second line of the Balmer series in the hydrogen spectrum is 4861 Å. Calculate the wavelength of the first line of the same series.
The figure below is the Energy level diagram for the Hydrogen atom. Study the transitions shown and answer the following question:
- State the type of spectrum obtained.
- Name the series of spectrum obtained.

