Advertisements
Advertisements
प्रश्न
The beaker is divided into two chambers A and B. The big circle represents solute and the small circles solvent.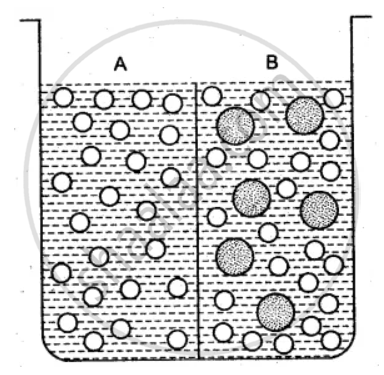
(i) What can you say about the size of the holes in the membrane, if it is to behave semi- permeably between these two?
(ii) Will the solvent molecules pass through the membrane from left to right, from right to left, in either direction or in both directions?
(iii) In which direction will there be a net movement of solvent molecules?
उत्तर
(i) The size of the holes in the membrane is large enough to allow only the solvent particles to pass through it. But solute particle cannot pass through it. Thus, the membrane acts as the semi-permeable.
(ii) Solvent molecules will pass through the membrane in both directions. Since solvent molecules are present on both the sides they will strike the semi-permeable membrane and pass through the same.
(iii) There is a net movement of solvent molecules from the place of its higher chemical potential to the place of its lower chemical potential, i.e., from right to left.
APPEARS IN
संबंधित प्रश्न
Differentiate between Diffusion and Osmosis (Definition)
Name the following:
The process by which raisins swell up when placed in a beaker of water.
Define the Cohesive and Adhesive forces.
Give Reasons for the following.
Salt and sugar are used in preserving food.
Given below is the figure of an experimental set up to demonstrate root pressure.
(i) Define root pressure.
(ii) What change would you observe in the water level after some time?
(iii) What role is being played by the root pressure in the given experiment?
(iv) Why the oil has been sprinkled on water?
Mention, if the following statement is True or False. If false rewrite the wrong statement in its correct form:
Cells that have lost their water content are said to be deplasmolysed.
Multiple Choice Question:
Plasma membrane controls:
