Advertisements
Advertisements
प्रश्न
The equation for the burning of octane is:
\[\ce{2C6H18 + 25O2 -> 16CO2 + 18H2O}\]
If the relative molecular mass of carbon dioxide is 44, what is the mass of carbon dioxide produced by burning two moles of octane?
उत्तर
1 mole of carbon dioxide has molecular mass = 44
16 moles of carbon dioxide has molecular mass = 44 × 16
= 704 g
संबंधित प्रश्न
Give balanced chemical equations for the following conversions A, B, and C:
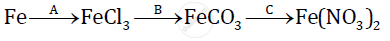
Calculate the relative molecular mass of Ammonium sulphate.
(use K = 39, Cl = 35.5, O = 16, C = 12, H = 1, Na = 23, N = 14, S= 32)
Concentrated nitric acid oxidizes phosphorous to phosphoric acid according to the following equation :
P + 5HNO3 → H3PO4 + H2O + 5NO2
What mass of nitric acid will be consumed at the same time?
If a crop of wheat removes 20 Kg of nitrogen per hectare of soil, what mass of the fertilizer calcium nitrate,Ca(NO3)2 would be required to replace nitrogen in 10 hectare field? (N = 14, O = 16, Ca = 40)
A metal M, forms a volatile chloride containing 65.5% Chlorine. If the density of the chloride relative to hydrogen is 162.5, find the molecular formula of the chloride. [M = 56, Cl = 35.5]
4.5 moles of calcium carbonate are reacted with dilute hydrochloric acid.
- Write the equation for the reaction.
- What is the mass of 4.5 moles of calcium carbonate? (Relative molecular mass of calcium carbonate is 100).
- What is the volume of carbon dioxide liberated at STP?
- What mass of calcium chloride is formed? (Relative molecular mass of calcium chloride is 111).
- How many moles of HCl are used in this reaction?
Calculate the volume of oxygen required for the complete combustion of 8.8 g of propane (C3H5).
(Atomic mass: C = 14, O = 16, H = 1, Molar Volume = 22.4 dm3 at STP.)
Calculate the relative molecular mass of:
Ammonium chloroplatinate (NH4)2 PtCl6
Calculate the relative molecular mass of:
CHCl3
Correct the statement, if required.
Under similar conditions of temperature and pressure, two volumes of hydrogen combined with two volumes of oxygen will give two volumes of water vapour.
