Advertisements
Advertisements
प्रश्न
The following statement is correct only under certain conditions. Rewrite the statement including the appropriate conditions.
Oxalic acid reacts with sulphuric acid to produce carbon monoxide and carbon dioxide.
उत्तर
Oxalic acid reacts with sulphuric acid to produce carbon monoxide.
APPEARS IN
संबंधित प्रश्न
Give a chemical test to distinguish between dilute sulphuric acid and conc. sulphuric acid.
Name the following:
Products obtained by treating ferrous sulphide with dilute sulphuric acid.
Name the following :
The precipitate obtained by treating aqueous lead nitrate with dilute sulphuric acid.
Give reason for the following:
When solution of sulphur dioxide is exposed to air, it gets converted to sulphuric acid.
Give reason for the following:
When concentrated sulphuric acid is added to blue crystalline copper sulphate, it turns powdery white.
How are the following conversion brought about? Give equation and condition:
Sucrose to sugar charcoal.
How are the following conversion brought about? Give equation and condition:
Oxalic acid to carbon monoxide.
Complete the following sentence, choosing the correct word from the given options.
"Concentrated sulphuric acid is used in the laboratory preparation of nitric acid and hydrochloric acid because it is ____________ in comparison to these two acids."
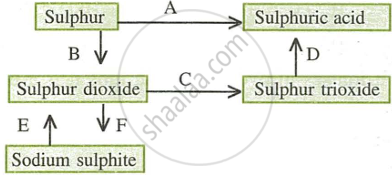
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
A, B, C and D summarize the properties of sulphuric acid depending on whether it is dilute or concentrated. Choose the property (A, B, C or D), depending on which is relevant to each of the preparations (i) to (ii).
A. Dilute acid (typical acid properties)
B. Non-volatile acid
C. Oxidizing agent
D. Dehydrating agent
(i) Preparation of hydrogen chloride.
(ii) Preparation of ethane from ethanol
(iii) Preparation of copper sulphate from copper oxide.
